- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
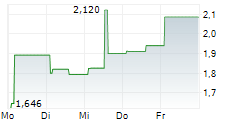
| Mo | Advicenne Granted Reimbursement for Likozam in France and Sees Thereby Its Debt Reduced by Nearly €6 Million | 189 | Business Wire | Likozam will be reimbursed to patients starting June 12, 2026 Official reimbursement ends the "Early Access" clawback The price obtained offsets the discounts owed under the early... ► Artikel lesen | |
| 21.05. | Advicenne Announces Changes to Its Governance Following Its Annual General Meeting on May 21, 2026 | 346 | Business Wire | Regulatory News:
Advicenne (Euronext Growth Paris FR0013296746 -ALDVI), a pharmaceutical company specializing in the development and marketing of innovativetreatments for people suffering from... ► Artikel lesen | |
| 05.05. | Advicenne Announces the Opening of a Safeguard Proceedings to Restructure Its Financial Liabilities and Finalize the Extension of Its Cash Runway | 490 | Business Wire | A proactive initiative to restructure the Company's debt A protective framework that allows for the continuation of commercial, clinical, and regulatory activities, as well as discussions... ► Artikel lesen | |
| 24.04. | ADVICENNE: Advicenne 2025 Universal Registration Document made available | 1 | Euronext | ||
| 24.04. | Advicenne 2025 Universal Registration Document Made Available | 353 | Business Wire | Regulatory News:
Advicenne (Euronext Growth Paris FR0013296746 ALDVI), a pharmaceutical company specializing in the development and marketing of innovative treatments for people suffering from... ► Artikel lesen | |
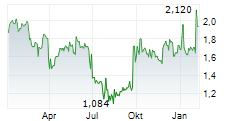
| 26.03. | Advicenne Reports Its 2025 Annual Financial Results | 3.205 | Business Wire | Product sales up 18.7% to €5.8 million
European end-market sales of Sibnayal rose by more than 71% to nearly €11 million
Royalties doubled to over €1.1 million
Cash position... ► Artikel lesen | |
| ADVICENNE Aktie jetzt für 0€ handeln | |||||
| 02.02. | Advicenne: Official Reimbursement of Sibnayal for the Treatment of dRTA in France | 1.415 | Business Wire | Regulatory News:
Advicenne (Euronext Growth Paris FR0013296746 ALDVI), a pharmaceutical company specializing in the development and marketing of innovative treatments for people suffering from... ► Artikel lesen | |
| 27.01. | Advicenne Receives Marketing Authorization and Reimbursement for Sibnayal in UAE | 424 | Business Wire | Regulatory News:
Advicenne (Euronext Growth Paris FR0013296746 ALDVI), a pharmaceutical company specializing in the development and marketing of innovative treatments for people suffering from... ► Artikel lesen | |
| 22.01. | Advicenne: 2025 Full Year Sales | 498 | Business Wire | 2025 sales totaled €5.73m, up 18.4%
Sibnayal sales rose 36.6% to €3.01m
2025 Royalties are estimated at €1m, double the amount for 2024
End-markets estimated sales of Sibnayal... ► Artikel lesen | |
| 20.01. | Advicenne: 2026 Financial Calendar | 440 | Business Wire | Regulatory News:
Advicenne(Euronext Growth Paris ALDVI FR0013296746), a specialty pharmaceutical company dedicated to the development and commercialization of innovative treatments for those suffering... ► Artikel lesen | |
| 19.01. | Advicenne's Sibnayal NDA For DRTA Tratement Accepted For Review By FDA | 2 | RTTNews | ||
| 19.01. | Advicenne: FDA Has Accepted for Review the Marketing Authorization Application for Sibnayal in the United States for the Treatment of dRTA | 342 | Business Wire | The US regulatory agency has set the target date for the final approval decision, known as the PDUFA date, as of September 3rd, 2026.
Regulatory News:
Advicenne (Euronext Growth Paris FR0013296746... ► Artikel lesen | |
| 12.01. | Advicenne Has Successfully Renewed the Marketing Authorization of Sibnayal in European Union | 332 | Business Wire | Regulatory News:
Advicenne (Euronext Growth Paris FR0013296746 ALDVI), a pharmaceutical company specializing in the development and marketing of innovative treatments for people suffering from... ► Artikel lesen | |
| 04.11.25 | Advicenne Has Submitted to US FDA the Registration Application for Sibnayal in dRTA Treatment | 523 | Business Wire | Regulatory News:
Advicenne (Euronext Growth Paris FR0013296746 ALDVI), a pharmaceutical company specializing in the development and marketing of innovative treatments for people suffering from... ► Artikel lesen | |
| 19.09.25 | Advicenne Reports First Half Financial Results as of June 30, 2025, and Updates on Its Activities | 588 | Business Wire | Sibnayal® end-market sales (Europe and Middle East) up 103% to €5.5 million
H1 products sales and royalties totaled €3.1 million, up 20%
Financial visibility extended to the fourth... ► Artikel lesen | |
| 26.08.25 | Advicenne Announces the Publication in Orphanet Journal of Rare Diseases of the Long-term European Study Results of dRTA Treatment Using ADV7103 | 431 | Business Wire | This extensive clinical dataset serves ADV7103 registration dossier in the United States of America, expected to be submitted in Q4 2025
Regulatory News:
Advicenne (Euronext Growth Paris... ► Artikel lesen | |
| 28.07.25 | Advicenne Receives Marketing Authorization and Reimbursement for Sibnayal in Saudi Arabia | 607 | Business Wire | Regulatory News:
Advicenne (Euronext Growth® FR0013296746 ALDVI), a pharmaceutical company specializing in the development and marketing of innovative treatments for people suffering from rare... ► Artikel lesen | |
| 25.07.25 | Advicenne Reports its H1 2025 Sales | 529 | Business Wire | Regulatory News:
Advicenne (Euronext Growth FR0013296746 ALDVI), a pharmaceutical company specializing in the development and marketing of innovative treatments for people suffering from rare kidney... ► Artikel lesen | |
| 21.07.25 | Advicenne Raises 2.6 Million Euros in Its Capital Increase and Extends Its Financial Visibility | 989 | Business Wire | Subscription of €2.6 million, representing 100% of the initial amount Financial horizon extended to the end of Q3 2026 thanks to concomitant financial restructuring Bpifrance now... ► Artikel lesen | |
| 02.07.25 | XFRA CAPITAL ADJUSTMENT INFORMATION - 02.07.2025 | 540 | Xetra Newsboard | Das Instrument 3MM FR0013296746 ADVICENNE (PROM.) EO-,20 EQUITY wird ex Kapitalmassnahme gehandelt am 02.07.2025 The instrument 3MM FR0013296746 ADVICENNE (PROM.) EO-,20 EQUITY is traded ex capital... ► Artikel lesen |
1 2 Weiter >>
21 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| PFIZER | 22,020 | -0,07 % | BARCLAYS stuft PFIZER INC auf 'Underweight' | LONDON (dpa-AFX Analyser) - Die britische Investmentbank Barclays hat die Einstufung für Pfizer mit einem Kursziel von 25 US-Dollar auf "Underweight" belassen. Angesichts der bisherigen Erfahrungen... ► Artikel lesen | |
| ABBVIE | 183,30 | +0,11 % | AbbVie Announces European Commission Approval of AQUIPTA (atogepant) for the Acute Treatment of Migraine in Adults | AQUIPTA (atogepant) is now approved in the European Union for the acute treatment of migraine in adults, with or without aura, and for the prophylaxis of migraine in adults who have at least... ► Artikel lesen | |
| HAEMATO | 13,000 | -1,52 % | M1 Kliniken schließt Verkauf von Haemato Pharm ab | Die M1 Kliniken AG hat den Verkauf der Haemato Pharm GmbH über ihre Mehrheitsbeteiligung Haemato AG an die Phoenix Group mit Wirkung zum Ablauf des 31. Januar 2026 abgeschlossen. Sämtliche Vollzugsbedingungen... ► Artikel lesen | |
| HALEON | 3,801 | -0,21 % | Haleon schließt Aktienrückkauf ab: Über 10 Millionen Aktien werden eingezogen | ||
| ACHIEVE LIFE SCIENCES | 4,480 | +0,90 % | Achieve Life Sciences Strengthens Board and Commercial Leadership with Three Senior Appointments from Team Previously at Verona Pharma | SEATTLE and VANCOUVER, British Columbia, May 12, 2026 (GLOBE NEWSWIRE) -- Achieve Life Sciences, Inc. (Achieve and the Company) (Nasdaq: ACHV), a late-stage specialty pharmaceutical company focused... ► Artikel lesen | |
| LIGAND PHARMACEUTICALS | 191,00 | -3,54 % | Orchestra BioMed Holdings, Inc.: Orchestra BioMed Receives $15 Million Investment from Ligand Under Previously Announced Strategic Financing Agreement | $15 million payment fulfills previously scheduled tranche under royalty-based financing agreement, bringing total capital received from Ligand to $40 million to date in exchange for tiered royalty... ► Artikel lesen | |
| ROYALTY PHARMA | 47,130 | -0,65 % | Royalty Pharma plc - 8-K, Current Report | ||
| EISAI | 20,430 | -0,92 % | Eisai Expects Revenue Of JPY 1 Trillion For 2028 | TOKYO (dpa-AFX) - Eisai Co., Limited (ESALY.PK, ESALF.PK, 4523.T, EII.SG), a Japanese pharmaceutical company, said on Monday that it expects revenue of JPY 1 trillion for fiscal 2028, helped... ► Artikel lesen | |
| TG THERAPEUTICS | 31,800 | -1,55 % | TG Therapeutics, Inc.: TG Therapeutics Announces Positive Topline Results from Phase 3 ENHANCE Trial | Phase 3 trial met its primary endpoint, demonstrating bioequivalent drug exposure between the currently approved BRIUMVI Day 1 and Day 15 initiation dosing and a new single infusion on Day 1 only... ► Artikel lesen | |
| DR REDDYS | 11,500 | 0,00 % | DR REDDYS LABORATORIES LTD - 20-F, Annual and transition report of foreign private issuers | ||
| BETTERLIFE PHARMA | 0,030 | 0,00 % | BetterLife Pharma Inc. - 20-F, Annual and transition report of foreign private issuers | ||
| ENTOURAGE HEALTH | - | - | Ontario Securities Commission: OSC bans WeedMD ex Carr, again | ||
| WAVE LIFE SCIENCES | 5,450 | +0,93 % | Wave Life Sciences Ltd.: Wave Life Sciences Announces Positive Update on RestorAATion-2 Trial: WVE-006 (GalNAc-RNA Editing) Achieves MZ-Like Phenotype Across Both Biweekly and Monthly Dosing | Data reinforce WVE-006's potential to address both lung and liver manifestations of AATD with a durable, convenient, and safe therapy capable of recapitulating the protective MZ-like phenotype with... ► Artikel lesen | |
| KYOWA KIRIN | 12,600 | -3,08 % | Kyowa Kirin: FDA Approves New Dosing Option for CRYSVITA (burosumab-twza) in Adults With XLH | Expanded dosing options support individualized treatment for adults with XLH PRINCETON, N.J., May 14, 2026 (GLOBE NEWSWIRE) -- Kyowa Kirin, Inc., a wholly owned subsidiary of Kyowa Kirin Co. Ltd.... ► Artikel lesen | |
| IGC PHARMA | 0,242 | -3,97 % | IGC Pharma, Inc.: IGC Pharma Reports First Quarter 2026 Financial Results with CALMA Phase 2 in the final stage, with 80% Enrollment | - Continued Advancement of CALMA Phase 2 Trial - POTOMAC, MD / ACCESS Newswire / May 19, 2026 / IGC Pharma, Inc. (NYSE American:IGC) ("IGC" or the "Company"), a clinical-stage biotechnology company... ► Artikel lesen |
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu ADVICENNE finden Sie auf Wallstreet Online.