| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
| 1,174 | 1,222 | 20:36 |
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 18:06 | Results of Aelis Farma Combined General Meeting of June 3, 2026 | 207 | Business Wire | Regulatory News:
Aelis Farma (ISIN: FR0014007ZB4 Ticker: AELIS), a clinical-stage biopharmaceutical company specializing in the development of treatments for brain and peripheral diseases involving... ► Artikel lesen | |
| 13.05. | Aelis Farma: Combined General Meeting of June 3, 2026: Availability of Preparatory Documents and Participation and Voting Procedures | 465 | Business Wire | Regulatory News:
Aelis Farma (ISIN: FR0014007ZB4 Ticker: AELIS), a clinical-stage biopharmaceutical company specializing in the development of treatments for brain and peripheral diseases involving... ► Artikel lesen | |
| AELIS FARMA Aktie jetzt für 0€ handeln | |||||
| 24.04. | Aelis Farma: Availability of the 2025 Universal Registration Document | 342 | Business Wire | Regulatory News:
Aelis Farma (ISIN: FR0014007ZB4 Ticker: AELIS), a clinical-stage biopharmaceutical company specializing in the development of treatments for brain and peripheral diseases involving... ► Artikel lesen | |
| 07.04. | Aelis Farma erhält 458.000 Euro für Adipositas-Forschung | 2 | Investing.com Deutsch | ||
| 07.04. | Aelis Farma receives €458,000 grant for obesity research | 1 | Investing.com | ||
| 07.04. | Aelis Farma Awarded a €458k Grant Under France 2030 in Nouvelle-Aquitaine to Support Development of Its Program in Obesity and Associated Metabolic Diseases | 310 | Business Wire | This non-dilutive funding, granted under the Projets d'Avenir Innovation (PAI) scheme, strengthens Aelis Farma's "metabolism" program and validates its innovation potential in a therapeutic area... ► Artikel lesen | |
| 30.03. | Aelis Farma Reports Its 2025 Annual Financial Results and Confirms Its 2026 Outlook | 597 | Business Wire | 2025 has been marked by several significant events: The achievement of key milestones for the Company's two drug candidates:
AEF0117: announcement of the final results of the Phase... ► Artikel lesen | |
| 21.03. | Aelis Farma startet Patientenrekrutierung für Studie zu Down-Syndrom-Medikament | 2 | Investing.com Deutsch | ||
| 21.03. | Aelis Farma begins recruiting for Down syndrome drug trial | 2 | Investing.com | ||
| 21.03. | Aelis Farma announces the successful start of the recruitment of the Phase 2B clinical trial with AEF0217 for the treatment of behavioral and cognitive impairments of people with Down syndrome (Trisomy 21) | 753 | Business Wire | AEF0217 is a first-in-class Signalling Specific inhibitor of the CB1 receptor (CB1-SSi) developed as a treatment for impairments in adaptive behaviours and cognition in neurodevelopmental disorders... ► Artikel lesen | |
| 26.01. | Aelis Farma Announces Its 2026 Financial Calendar | 300 | Business Wire | Regulatory News:
Aelis Farma (ISIN: FR0014007ZB4 Ticker: AELIS, PEA-PME eligible), a clinical-stage biopharmaceutical company specializing in the development of treatments for brain and peripheral... ► Artikel lesen | |
| 12.01. | Aelis Farma Receives a Positive Opinion From EMA Pediatric Committee on the Pediatric Investigation Plan for AEF0217 in Down Syndrome | 388 | Business Wire | The EMA's Pediatric Committee (PDCO) has delivered a favorable consensus opinion on the Pediatric Investigational Plan (PIP) of Aelis Farma's first-in-class drug candidate AEF0217 for the treatment... ► Artikel lesen | |
| 02.12.25 | Aelis Farma Obtains Regulatory Approval of the Phase 2B Trial With AEF0217 in People With Down Syndrome | 465 | Business Wire | AEF0217 is a first-in-class CB1 receptor Signalling Specific inhibitor (CB1-SSi) developed as a potential first pharmacological treatment for impairments in adaptive behaviours and cognition... ► Artikel lesen | |
| 25.09.25 | Aelis Farma: Availability of the 2025 Half-Year Financial Report | 421 | Business Wire | Regulatory News:
Aelis Farma (ISIN: FR0014007ZB4 Ticker: AELIS), a clinical-stage biopharmaceutical company specialising in the development of treatments for brain and peripheral diseases involving... ► Artikel lesen | |
| 22.09.25 | Aelis Farma Reports 2025 Half-Year Financial Results and Presents Progress and Development Outlook | 615 | Business Wire | The first half of 2025 was marked by: The publication of the final analysis of the pioneering Phase 2b clinical trial in cannabis use disorders (CUD) with the CB1-SSi AEF0117, which further... ► Artikel lesen | |
| 24.06.25 | Aelis Farma: Availability of the Description of the Share Buyback Program | 462 | Business Wire | Regulatory News:
Aelis Farma (ISIN: FR0014007ZB4 Ticker: AELIS), a clinical-stage biopharmaceutical company specializing in the development of treatments for brain and peripheral diseases involving... ► Artikel lesen |
16 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| QIAGEN | 32,630 | +5,34 % | DEUTSCHE BANK RESEARCH stuft QIAGEN NV auf 'Buy' | FRANKFURT (dpa-AFX Analyser) - Deutsche Bank Research hat die Einstufung für Qiagen mit einem Kursziel von 43 US-Dollar auf "Buy" belassen. Das Interesse der Investoren habe vor allem dem Tuberkulosetest... ► Artikel lesen | |
| RECURSION PHARMACEUTICALS | 3,850 | +6,65 % | Recursion Pharmaceuticals: Recursion Reports Grant of Inducement Awards as Permitted by the Nasdaq Listing Rules | ||
| BEAM THERAPEUTICS | 32,790 | +13,23 % | Beam Therapeutics Announces Compelling Updated Clinical Data from the Ongoing Phase 1/2 Trial of BEAM-302 in Alpha-1 Antitrypsin Deficiency (AATD) to Support Advancement to Pivotal Development | Treatment with 60 mg of BEAM-302 Led to Mean Steady-state Total AAT Level of 16.1 µM and All Patients Consistently and Durably Above the 11 µM Protective AAT Threshold with up to 12 Months of Follow-up... ► Artikel lesen | |
| PRAXIS PRECISION MEDICINES | 271,00 | +5,01 % | Praxis Precision Medicines, Inc.: Praxis Precision Medicines Provides Vormatrigine Program Update | POWER1 Study in highly refractory patients with focal onset seizures did not meet its primary success measure Secondary measure, the 50% response rate, was met and seizure reduction during the second... ► Artikel lesen | |
| ERASCA | 13,370 | -0,82 % | Erasca, Inc.: Erasca Reports First Quarter 2026 Business Updates and Financial Results | Robust monotherapy efficacy and generally well-tolerated safety results observed during dose escalation for ERAS-0015 in both KRAS G12X NSCLC and PDAC reinforce best-in-class potential across RAS-targeted... ► Artikel lesen | |
| SUMMIT THERAPEUTICS | 14,505 | -2,39 % | All You Need to Know About Summit Therapeutics (SMMT) Rating Upgrade to Buy | ||
| VERA THERAPEUTICS | 36,800 | +17,68 % | Vera Therapeutics Announces Alignment with U.S. FDA on Earlier ORIGIN Phase 3 Analysis to Support Potential Full Approval for Atacicept in Adults with IgA Nephropathy | Prescription Drug User Fee Act (PDUFA) date of July 7, 2026 for Biologics License Application (BLA) for accelerated approval of atacicept in patients with IgA Nephropathy (IgAN)Revised ORIGIN 3 eGFR... ► Artikel lesen | |
| ABSCI | 7,040 | +10,87 % | BTIG initiates Absci stock with buy rating on AI drug platform | ||
| APOGEE THERAPEUTICS | 88,43 | +12,35 % | Weekly Buzz: AstraZeneca, JNJ Get FDA Nod; Eli Lilly To Acquire Curevo; APGE Advances Zumilokibart | NEW BRUNSWICK (dpa-AFX) - This week in the biotech space, there were regulatory approvals across the U.S. and Europe, collaborations, acquisitions, and positive data readouts spanning multiple... ► Artikel lesen | |
| JADE BIOSCIENCES | 18,160 | +2,31 % | Jade Biosciences Announces Positive Interim Phase 1 Results for JADE101 Demonstrating Potential Best-in-Class Profile and Every-12-Week Dosing in IgA Nephropathy | ~70% IgA reductions sustained at 12 weeks after a single dose of JADE101>70% IgA reductions projected at steady state with a single subcutaneous maintenance injection every 12 weeksIgA-lowering effect... ► Artikel lesen | |
| INDAPTUS THERAPEUTICS | 3,730 | +68,78 % | Indaptus Therapeutics, Inc. - 10-Q, Quarterly Report | ||
| COGENT BIOSCIENCES | 32,950 | +0,58 % | Cogent Biosciences, Inc.: Cogent Biosciences Announces Detailed Clinical Data from PEAK Phase 3 Trial with Bezuclastinib in Combination with Sunitinib in Gastrointestinal Stromal Tumors (GIST) at 2026 American Society of Clinical Oncology (ASCO) ... | Bezuclastinib combination is first treatment ever to demonstrate statistically significant advantage against active comparator in GIST patients; median PFS of 16.5 months versus 9.2 months (HR=0.50... ► Artikel lesen | |
| MINERALYS THERAPEUTICS | 25,320 | +1,04 % | Mineralys Therapeutics, Inc.: Mineralys Therapeutics Announces Repurchase of Royalty Obligations in Tanabe License Agreement and Concurrent Financing | - Repurchase of lorundrostat royalty obligation in Tanabe license agreement for $200 million upfront and up to $100 million once certain commercial milestones are met - - $500 million committed... ► Artikel lesen | |
| RELAY THERAPEUTICS | 14,350 | -8,60 % | Relay Therapeutics Shares Jump 18% | ||
| TYRA BIOSCIENCES | 27,780 | -3,17 % | Tyra Biosciences, Inc. - 8-K, Current Report |
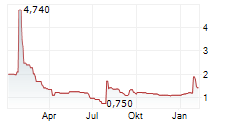
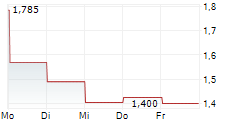
Das Unternehmen AELIS FARMA SAS kann der Branche Biotechnologie zugeordnet werden. Mit einer aktuellen Kursveränderung von +0,046 (+3,83 %) liegt der Kurs bei 1,246 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu AELIS FARMA SAS finden Sie auf Wallstreet Online.