| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
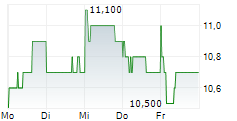
| 12,665 | 12,835 | 13:04 | |
| 12,710 | 12,910 | 22.05. |
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| Fr | HSCEI Adds HANSOH PHARMA and AKESO, Removes SUNNY OPTICAL and HAIER SMARTHOME in Quarterly Review | 7 | AASTOCKS | ||
| 15.05. | Akeso, Inc.: Phase II Data from a Randomized Double-Blind Trial of Ligufalimab (Anti-CD47) Combination Therapy in Frontline AML Published at EHA 2026 | 286 | PR Newswire | HONG KONG, May 15, 2026 /PRNewswire/ -- Akeso, Inc. (9926.HK) ("Akeso" or the "Company") today announced that compelling results from its randomized, double-blind... ► Artikel lesen | |
| 06.05. | JPM: AKESO Lung Cancer Trial Final Analysis Likely to Meet Target; Rating Overweight | 4 | AASTOCKS | ||
| 01.05. | Summit Therapeutics falls after update on Akeso-partnered lung cancer trial | 41 | Seeking Alpha | ||
| 29.04. | AKESO (09926): NOTIFICATION LETTER AND REPLY FORM TO NON-REGISTERED HOLDERS - REMINDER LETTER REGARDING THE ARRANGEMENT OF ELECTRONIC DISSEMINATION OF ... | - | HKEx | ||
| 29.04. | AKESO (09926): NOTIFICATION LETTER AND REPLY FORM TO REGISTERED SHAREHOLDERS - REMINDER LETTER REGARDING THE ARRANGEMENT OF ELECTRONIC DISSEMINATION OF ... | 1 | HKEx | ||
| 29.04. | AKESO (09926): 2025 ENVIRONMENTAL, SOCIAL AND GOVERNANCE REPORT | - | HKEx | ||
| AKESO Aktie jetzt für 0€ handeln | |||||
| 29.04. | AKESO (09926): 2025 ANNUAL REPORT | - | HKEx | ||
| 28.04. | BofAS: Akeso Cadonilimab Records MoM Growth Against Seasonality | 2 | AASTOCKS | ||
| 21.04. | Akeso, Inc.: Cadonilimab Combination Demonstrates Promising Survival Benefit in Locally Advanced Pancreatic Cancer: Phase II COMPASSION-26 Data Presented at AACR 2026 | 129 | PR Newswire | HONG KONG, April 20, 2026 /PRNewswire/ -- Akeso, Inc. (9926.HK) ("Akeso" or the "Company") today announced that positive Phase II results from the COMPASSION-26... ► Artikel lesen | |
| 27.03. | Akeso, Inc.: Akeso Reports Full-Year 2025 Financial Results | 616 | PR Newswire | HONG KONG, March 26, 2026 /PRNewswire/ -- Akeso, Inc. (9926.HK) ("Akeso" or the "Company") announced its 2025 annual results, highlighting a year of comprehensive... ► Artikel lesen | |
| 26.03. | AKESO (09926): ANNUAL RESULTS ANNOUNCEMENT FOR THE YEAR ENDED DECEMBER 31, 2025 | 2 | HKEx | ||
| 25.03. | Akeso, Inc.: Akeso Advances "IO 2.0 + ADC 2.0" Strategy with Phase II Initiation of Novel ADCs Combined with Ivonescimab and Cadonilimab | 221 | PR Newswire | HONG KONG, March 24, 2026 /PRNewswire/ -- Akeso, Inc. (9926.HK) ("Akeso" or the "Company") announced today that it has received clearance from the Center for Drug... ► Artikel lesen | |
| 16.03. | AKESO (09926): DATE OF BOARD MEETING | 3 | HKEx | ||
| 16.03. | Akeso, Inc.: Akeso Announces Global First-in-Class Trispecific Antibody AK150 Enters Clinical Trials: A Triple-Target Approach to Overcome Immunotherapy Resistance | 276 | PR Newswire | HONG KONG, March 15, 2026 /PRNewswire/ -- Akeso, Inc. (9926.HK) ("Akeso" or the "Company") announced today that its proprietary first-in-class trispecific antibody... ► Artikel lesen | |
| 05.03. | Akeso, Inc.: Cadonilimab Achieves 100% 24-Month OS in Complete Responders in R/M Cervical Cancer Based on Long-Term Phase II Results | 214 | PR Newswire | HONG KONG, March 4, 2026 /PRNewswire/ -- Akeso, Inc. (9926.HK) ("Akeso" or the "Company") announced that the latest long-term survival analysis data from the China... ► Artikel lesen | |
| 04.03. | Akeso, Inc.: Akeso and INOVIO Announce Clinical Collaboration to Advance Novel Combination Therapy for Glioblastoma (GBM) | 1.030 | PR Newswire | Collaboration will evaluate Akeso's cadonilimab in combination with INOVIO's INO-5412 (INO-5401 plus INO-9012) as a potential treatment for glioblastoma (GBM)... ► Artikel lesen | |
| 04.03. | INOVIO Pharmaceuticals, Inc.: INOVIO and Akeso Announce Clinical Collaboration to Advance Novel Combination Therapy for Glioblastoma (GBM) | 514 | PR Newswire | Collaboration will evaluate INOVIO's INO-5412 (INO-5401 plus INO-9012) in combination with Akeso's cadonilimab as a potential treatment for glioblastoma (GBM), the most common and aggressive... ► Artikel lesen | |
| 03.02. | AKESO (09926): VOLUNTARY ANNOUNCEMENT - AKESO GRANTED EXCLUSIVE COMMERCIALIZATION RIGHTS FOR EBRONUCIMAB INJECTION TO JUMPCAN PHARMACEUTICAL | 6 | HKEx | ||
| 26.01. | AKESO (09926): CONTINUING CONNECTED TRANSACTION FURTHER INFORMATION IN RELATION TO THE CONTRACT MANUFACTURING ARRANGEMENT | 5 | HKEx |
1 2 3 Weiter >>
45 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BIONTECH | 79,35 | +1,67 % | BioNTech-Aktie: Gap-Close und bereit für +24%? | Die BioNTech-Aktie hat den zuletzt markanten Unterstützungsbereich um 76,40 € angelaufen und eine Reaktion gezeigt. Heute mit Up-Gap gestartet, könnte sich eine zeitnahe Schließung dieser Eröffnungslücke... ► Artikel lesen | |
| EVOTEC | 5,230 | +2,95 % | KAUFKURSE bei Renk und Desert Gold! SCHOCK bei der Evotec Aktie! | Während der Goldpreis schwächelt, befindet sich die Aktie von Desert Gold in einem sauberen Aufwärtstrend. Und geht es nach Analysten, ist eine Vervielfachung möglich. Denn Desert Gold wird in wenigen... ► Artikel lesen | |
| MEDIGENE | 0,037 | +18,59 % | MEDIGENE AG im Fokus - ruhige Einordnung der Schwäche | ||
| QIAGEN | 30,290 | +0,65 % | QIAGEN N.V.: QIAGEN to advance AI-driven drug discovery with graph-based AI and curated bioinformatics knowledge with NVIDIA | Collaboration integrates NVIDIA accelerated computing and the NVIDIA BioNeMo platform with QIAGEN Digital Insights' curated biomedical knowledge bases Goal to advance how disease mechanisms... ► Artikel lesen | |
| MODERNA | 40,375 | -0,05 % | Moderna Flu Shot Heads To FDA Advisory Panel | ||
| PAION | 0,072 | -25,52 % | XFRA MISTRADE ANTRAG IN ISIN DE000A3E5EG5 WURDE STATTGEGEBEN | Dem Mistrade-Antrag wurde stattgegeben. Folgende Geschaefte in der ISIN DE000A3E5EG5, Wertpapier-Name: PAION AG INH O.N., wurden aufgehoben:ISIN Datum Zeit Volumen PreisDE000A3E5EG5 02.02.2026 08:13:40 300 0... ► Artikel lesen | |
| VALNEVA | 2,490 | +0,81 % | Valneva Aktie: Prognose kassiert - die Börse schaut woanders hin | Valneva hat das erste Quartal 2026 mit einem deutlichen Umsatzrückgang abgeschlossen und die Jahresprognose gesenkt. Der Aktienkurs reagierte nicht entscheidend, denn der Markt richtet seinen Blick... ► Artikel lesen | |
| AMGEN | 292,50 | +0,05 % | Mesoblast im Sog der Big Player: Wie Merck, Novartis und Johnson & Johnson den Zelltherapie-Markt vorbereiten | ||
| NOVAVAX | 7,930 | 0,00 % | Novavax, Inc.: Novavax Reports First Quarter 2026 Financial Results and Operational Highlights | Total revenue of $140 million in the first quarter of 2026
Pfizer agreement announced in January for non-exclusive license to utilize Matrix-M® in two infectious... ► Artikel lesen | |
| STRYKER | 272,50 | -0,07 % | Stryker Buys Amplitude Vascular Systems For Upto $835 Mln To Expand Vascular Treatment Business | WASHINGTON (dpa-AFX) - Tuesday, BioStar Capital announced that Amplitude Vascular Systems, Inc., a medical technology company, has been acquired by Stryker Corporation (SYK) at a purchase price... ► Artikel lesen | |
| BIOGEN | 166,64 | -0,22 % | XETR DELETION OF INSTRUMENTS FROM XETRA - 22.05.2026 | The following instruments on XETRA do have their last trading day on 22.05.2026.Die folgenden Instrumente auf XETRA haben ihren letzten Handelstag am 22.05.2026..ISIN Name US09062X1037 Biogen Inc.US2521311074 DexCom... ► Artikel lesen | |
| BIOFRONTERA | 2,390 | -1,65 % | Biofrontera stock rating cut to Speculative Buy by Benchmark | ||
| HEIDELBERG PHARMA | 2,740 | -0,72 % | EQS-HV: Heidelberg Pharma AG: Bekanntmachung der Einberufung zur Hauptversammlung am 23.06.2026 in Ladenburg mit dem Ziel der europaweiten Verbreitung gemäß §121 AktG | EQS-News: Heidelberg Pharma AG
/ Bekanntmachung der Einberufung zur Hauptversammlung
Heidelberg Pharma AG: Bekanntmachung der Einberufung zur Hauptversammlung am 23.06.2026 in... ► Artikel lesen | |
| ILLUMINA | 124,52 | +0,10 % | Illumina adds former Labcorp CEO to board of directors | ||
| SUMMIT THERAPEUTICS | 16,620 | 0,00 % | This Summit Therapeutics Analyst Is No Longer Bullish; Here Are Top 2 Downgrades For Friday |
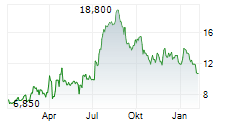
Das Unternehmen AKESO INC kann der Branche Biotechnologie zugeordnet werden. Mit einer aktuellen Kursveränderung von 0,000 (0,00 %) liegt der Kurs bei 12,715 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu AKESO INC finden Sie auf Wallstreet Online.