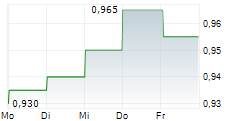
| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
| 0,965 | 1,000 | 13:04 | |
| 0,960 | 0,995 | 15.05. |
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| Mi | ALPHAMAB-B (09966): VOLUNTARY ANNOUNCEMENT - FIRST PATIENT DOSED IN A PHASE II CLINICAL STUDY OF JSKN033 AS FIRST-LINE TREATMENT OF ADVANCED CERVICAL ... | 2 | HKEx | ||
| 28.04. | ALPHAMAB-B (09966): 2025 ENVIRONMENTAL, SOCIAL AND GOVERNANCE REPORT | 1 | HKEx | ||
| ALPHAMAB ONCOLOGY Aktie jetzt für 0€ handeln | |||||
| 28.04. | ALPHAMAB-B (09966): 2025 ANNUAL REPORT | - | HKEx | ||
| 23.04. | ALPHAMAB-B (09966): GRANT OF AWARD SHARES UNDER THE RESTRICTED SHARE AWARD SCHEME | - | HKEx | ||
| 22.04. | ALPHAMAB-B (09966): WAIVER FROM STRICT COMPLIANCE WITH RULES 3.28 AND 8.17 OF THE LISTING RULES | 1 | HKEx | ||
| 22.04. | Alphamab Oncology-B KN026 Neoadjuvant Therapy Selected for Presentation at 2026 ASCO Annual Meeting | 2 | AASTOCKS | ||
| 22.04. | ALPHAMAB-B (09966): VOLUNTARY ANNOUNCEMENT - SELECTION OF RESULTS OF PHASE III CLINICAL STUDY OF KN026 FOR NEOADJUVANT TREATMENT OF BC FOR LBA ORAL SESSION ... | 1 | HKEx | ||
| 08.04. | ALPHAMAB-B (09966): (1) CHANGE OF JOINT COMPANY SECRETARY AND AUTHORIZED REPRESENTATIVE; AND (2) WAIVER FROM STRICT COMPLIANCE WITH RULES 3.28 AND 8.17 ... | - | HKEx | ||
| 31.03. | ALPHAMAB-B (09966): VOLUNTARY ANNOUNCEMENT - A PHASE III CLINICAL STUDY OF KN026 FOR NEOADJUVANT TREATMENT OF HER2+ BC MET PRIMARY ENDPOINT | - | HKEx | ||
| 25.03. | ALPHAMAB-B (09966): ANNUAL RESULTS ANNOUNCEMENT FOR THE YEAR ENDED DECEMBER 31, 2025 | - | HKEx | ||
| 24.03. | ALPHAMAB-B (09966): VOLUNTARY ANNOUNCEMENT - FIRST PATIENT DOSED IN PHASE III CLINICAL STUDY OF KN026 FOR ADJUVANT TREATMENT OF HER2+ BC | 1 | HKEx | ||
| 20.03. | ALPHAMAB-B (09966): VOLUNTARY ANNOUNCEMENT - FIRST PATIENT DOSED IN A PHASE III CLINICAL STUDY OF JSKN016 FOR THE TREATMENT OF TNBC | 2 | HKEx | ||
| 18.03. | ALPHAMAB-B (09966): VOLUNTARY ANNOUNCEMENT - APPOINTMENT OF CHIEF FINANCIAL OFFICER | 2 | HKEx | ||
| 16.03. | ALPHAMAB-B (09966): VOLUNTARY ANNOUNCEMENT - FIRST PATIENT DOSED IN A PHASE I CLINICAL STUDY OF JSKN027 | 1 | HKEx | ||
| 13.03. | ALPHAMAB-B (09966): VOLUNTARY ANNOUNCEMENT - IND APPLICATION FOR JSKN021 WAS OFFICIALLY ACCEPTED BY THE CDE | 1 | HKEx | ||
| 11.03. | ALPHAMAB-B (09966): DATE OF BOARD MEETING | 1 | HKEx | ||
| 11.03. | ALPHAMAB-B (09966): VOLUNTARY ANNOUNCEMENT - APPOINTMENT OF CHIEF TECHNOLOGY OFFICER | 2 | HKEx | ||
| 09.01. | ALPHAMAB-B (09966): VOLUNTARY ANNOUNCEMENT - NDA FOR FIRST-LINE TREATMENT OF BTC OF KN035 WAS ACCEPTED BY THE NMPA | 1 | HKEx | ||
| 31.12.25 | ALPHAMAB-B (09966): NEXT DAY DISCLOSURE RETURN | - | HKEx | ||
| 29.12.25 | ALPHAMAB-B (09966): VOLUNTARY ANNOUNCEMENT - IND APPLICATION FOR A PHASE II CLINICAL TRIAL OF JSKN033 WAS OFFICIALLY ACCEPTED BY THE CDE | 1 | HKEx |
1 2 Weiter >>
24 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BIONTECH | 76,40 | -2,98 % | BioNTech-Krise vs. Emyria-Durchbruch? Warum Daten jetzt die Forschung schlagen | ||
| EVOTEC | 4,618 | -1,74 % | KURSRUTSCH bei Nel ASA und Evotec! KURSCHANCE bei HPQ Silicon! | Steht die Aktie von HPQ Silicon vor einer Neubewertung? Dafür gibt es jedenfalls gute Gründe. Die Hochleistungsbatterien haben erneut überzeugt. Damit könnte für Anwendungen in Drohnen, Verteidigung... ► Artikel lesen | |
| MEDIGENE | 0,032 | -9,04 % | PTA-Adhoc: Medigene AG: Widerruf der Zulassung der Aktien der Medigene AG zum Handel im regulierten Markt | DJ PTA-Adhoc: Medigene AG: Widerruf der Zulassung der Aktien der Medigene AG zum Handel im regulierten Markt
Veröffentlichung von Insiderinformationen gemäß Artikel 17 MAR Schlagwort(e): Sonstiges... ► Artikel lesen | |
| QIAGEN | 28,465 | -0,44 % | Qiagen erhöht Dividende und stellt weitere Aktienrückkäufe in Aussicht | DJ Qiagen erhöht Dividende und stellt weitere Aktienrückkäufe in Aussicht
DOW JONES--Die Aktionäre von Qiagen können sich über eine höhere Dividende freuen. Wie es in der Einladung zur Hauptversammlung... ► Artikel lesen | |
| MODERNA | 42,000 | -0,52 % | Moderna-Aktie steigt weiter: Hantavirus im Fokus | Die Sorge rund um das Hantavirus wächst: Auf dem Kreuzfahrtschiff "Hondius" wurden mehrere Infektionen mit der seltenen südamerikanischen Andes-Variante registriert, drei Personen starben. Trotz des... ► Artikel lesen | |
| PAION | 0,073 | -24,48 % | XFRA MISTRADE ANTRAG IN ISIN DE000A3E5EG5 WURDE STATTGEGEBEN | Dem Mistrade-Antrag wurde stattgegeben. Folgende Geschaefte in der ISIN DE000A3E5EG5, Wertpapier-Name: PAION AG INH O.N., wurden aufgehoben:ISIN Datum Zeit Volumen PreisDE000A3E5EG5 02.02.2026 08:13:40 300 0... ► Artikel lesen | |
| VALNEVA | 2,540 | +0,95 % | Valneva senkt Prognose und baut Stellen ab | Beim Impfstoffspezialisten Valneva SE häufen sich im ersten Quartal 2026 die Belastungen: Der Nettoverlust weitete sich auf 32,1 Millionen Euro nach 9,2 Millionen Euro im Vorjahr aus, das bereinigte... ► Artikel lesen | |
| AMGEN | 282,15 | +0,46 % | Dividendenbekanntmachungen (15.05.2026) | Unternehmen ISIN-Code Dividende (Währung) Dividende (EUR) A10 NETWORKS INC US0021211018 0,06 USD 0,0514 EUR AF GRUPPEN ASA NO0003078107 6,5 NOK 0,6029 EUR AGCO CORPORATION US0010841023 0... ► Artikel lesen | |
| NOVAVAX | 7,800 | 0,00 % | Novavax Stock Is Red Hot but the Smart Money Is Slamming on the Brakes | ||
| STRYKER | 264,60 | +0,23 % | General Dynamics unit wins $229.65M Army deal for 50 Stryker A1 vehicles | ||
| BIOGEN | 165,76 | -0,16 % | Biogen Inc.: Biogen Completes Acquisition of Apellis Pharmaceuticals | CAMBRIDGE, Mass., May 14, 2026 (GLOBE NEWSWIRE) -- Biogen Inc. (Nasdaq: BIIB) today announced the successful completion of the acquisition of Apellis Pharmaceuticals, Inc. (Nasdaq: APLS). Apellis... ► Artikel lesen | |
| BIOFRONTERA | 2,250 | -4,26 % | Biofrontera GAAP EPS of -$0.41 misses by $0.33, revenue of $10.1M misses by $0.4M | ||
| HEIDELBERG PHARMA | 2,740 | -0,72 % | EQS-HV: Heidelberg Pharma AG: Bekanntmachung der Einberufung zur Hauptversammlung am 23.06.2026 in Ladenburg mit dem Ziel der europaweiten Verbreitung gemäß §121 AktG | EQS-News: Heidelberg Pharma AG
/ Bekanntmachung der Einberufung zur Hauptversammlung
Heidelberg Pharma AG: Bekanntmachung der Einberufung zur Hauptversammlung am 23.06.2026 in... ► Artikel lesen | |
| ILLUMINA | 121,00 | -1,37 % | RBC Capital assumes Illumina stock coverage with outperform rating | ||
| TANGO THERAPEUTICS | 20,560 | 0,00 % | Tango Therapeutics, Inc.: Tango Therapeutics Reports First Quarter 2026 Financial Results and Provides Business Highlights | First clinical data from PRMT5/RAS(ON) combination trial to be presented in 2026 Cash position of $380 million as of March 31, 2026, with runway into 2028 beyond anticipated key data inflection points... ► Artikel lesen |
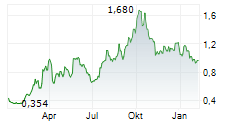
ALPHAMAB ONCOLOGY gehört der Branche Biotechnologie an. Die relative Kursveränderung beträgt aktuell -0,52 % bei einem Aktienkurs von 0,960 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu ALPHAMAB ONCOLOGY finden Sie auf Wallstreet Online.