| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
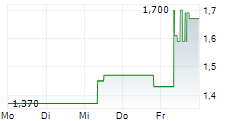
| 1,690 | 1,750 | 13:04 | |
| 1,680 | 1,750 | 22.05. |
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 06.05. | ASCLETIS-B to Present Multiple Projects in Poster Sessions at European Congress on Obesity | 1 | AASTOCKS | ||
| ASCLETIS PHARMA Aktie jetzt für 0€ handeln | |||||
| 06.05. | ASCLETIS-B (01672): VOLUNTARY ANNOUNCEMENT - ASCLETIS TO PRESENT DATA ON MULTIPLE PROGRAMS AT THE 33RD EUROPEAN CONGRESS ON OBESITY (ECO 2026) | 1 | HKEx | ||
| 30.04. | ASCLETIS-B to Present Preclinical Data of Weight-loss Candidate ASC39 at ADA Conference | 3 | AASTOCKS | ||
| 30.04. | ASCLETIS-B (01672): VOLUNTARY ANNOUNCEMENT - ASCLETIS TO PRESENT DATA ON MULTIPLE PROGRAMS AT THE AMERICAN DIABETES ASSOCIATION'S 2026 SCIENTIFIC SESSIONS | 1 | HKEx | ||
| 29.04. | ASCLETIS-B (01672): ANNUAL REPORT 2025 | 3 | HKEx | ||
| 27.04. | Ascletis Pharma-B Completes Enrollment in US Phase II Study of Oral Small Molecule GLP-1R Agonist ASC30 | 1 | AASTOCKS | ||
| 27.04. | ASCLETIS-B (01672): VOLUNTARY ANNOUNCEMENT - ASCLETIS COMPLETES ENROLLMENT IN U.S. PHASE II STUDY OF ASC30, AN ORAL SMALL MOLECULE GLP-1R AGONIST, FOR ... | 2 | HKEx | ||
| 07.04. | Ascletis Pharma Inc.: Ascletis Announces Fixed-Dose Combination of ASC30, Once-Daily Oral Small Molecule GLP-1R Agonist, and ASC39, Once-Daily Oral Small Molecule Amylin-Selective Amylin Receptor Agonist, for Clinical Development | 138 | PR Newswire | -Fixed-dose combination of ASC30 and ASC39 (ASC30_39 FDC) tablets, dosed orally in dogs, demonstrated comparable pharmacokinetics to those observed in their respective... ► Artikel lesen | |
| 07.04. | ASCLETIS-B (01672): VOLUNTARY ANNOUNCEMENT - ASCLETIS ANNOUNCES FIXED-DOSE COMBINATION OF ASC30, ONCE-DAILY ORAL SMALL MOLECULE GLP-1R AGONIST, AND ASC39, ... | 1 | HKEx | ||
| 31.03. | ASCLETIS-B (01672): ANNUAL RESULTS ANNOUNCEMENT FOR THE YEAR ENDED DECEMBER 31, 2025 AND CHANGE IN THE USE OF PROCEEDS | 1 | HKEx | ||
| 18.03. | ASCLETIS-B (01672): NOTICE OF BOARD MEETING | 1 | HKEx | ||
| 17.03. | ASCLETIS-B (01672): VOLUNTARY ANNOUNCEMENT - ORAL SMALL MOLECULE AMYLIN RECEPTOR AGONIST ASC39 DEMONSTRATED ELORALINTIDE-LIKE AMYLIN SELECTIVITY AND EFFICACY ... | 1 | HKEx | ||
| 10.03. | Ascletis eyes quarterly GLP-1 dosing following phase 2 obesity readout | 1 | FierceBiotech | ||
| 10.03. | Ascletis Pharma Inc.: Ascletis Announces Positive Topline Results from U.S. Phase II, 24-Week Study for Its Ultra-Long-Acting Subcutaneous Depot Formulations of Small Molecule GLP-1R Agonist ASC30 for Obesity | 666 | PR Newswire | - ASC30 subcutaneous (SQ) depot formulation achieved statistically significant and clinically meaningful placebo-adjusted mean weight loss of 7.5% at week 16... ► Artikel lesen | |
| 10.03. | ASCLETIS-B (01672): VOLUNTARY ANNOUNCEMENT - ASCLETIS ANNOUNCES POSITIVE TOPLINE RESULTS FROM U.S. PHASE II, 24-WEEK STUDY FOR ITS ULTRA-LONG-ACTING SUBCUTANEOUS ... | 1 | HKEx | ||
| 11.02. | ASCLETIS-B (01672): VOLUNTARY ANNOUNCEMENT - ASCLETIS SELECTS ORAL AMYLIN RECEPTOR PEPTIDE AGONIST, ASC36, FOR CLINICAL DEVELOPMENT | 2 | HKEx | ||
| 10.02. | ASCLETIS-B (01672): NEXT DAY DISCLOSURE RETURN | 4 | HKEx | ||
| 10.02. | ASCLETIS-B (01672): COMPLETION OF PLACING OF NEW SHARES UNDER THE GENERAL MANDATE | 1 | HKEx | ||
| 06.02. | Ascletis Pharma Inc.: GIC Invests US$100M in Ascletis Pharma (1672. HK): Anchoring Long-Term Capital in China's Core Innovative Drug Assets | 547 | JCN Newswire | HONG KONG, Feb 6, 2026 - (ACN Newswire) - According to disclosure of interests filed with the Hong Kong Stock Exchange (HKEX), the Government of Singapore Investment Corporation (GIC) has acquired an... ► Artikel lesen | |
| 06.02. | Ascletis Pharma Inc.: GIC Private Limited initiated a stake in Ascletis Pharma (01672) by 64,128,000 shares at a price of HKD 12.18 per share | 411 | JCN Newswire | HONG KONG, Feb 6, 2026 - (ACN Newswire) - According to the disclosure of interests information released by the Hong Kong Stock Exchange, on 5 February, GIC Private Limited made its first equity investment... ► Artikel lesen |
1 2 3 Weiter >>
59 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BIONTECH | 79,35 | +1,67 % | Übernahme Kandidaten für 2026! Heiß wird es im LifeScience-Sektor bei Evotec, BioNxt, BioNTech und Formycon! | Die Börse hat sich in den letzten Monaten primär auf HighTech und Rüstung konzentriert. Der gesamte BioTech-Sektor wurde damit links liegen gelassen. Kurzfristig mag diese Strategie für Investoren aufgegangen... ► Artikel lesen | |
| EVOTEC | 5,230 | +2,95 % | Shortseller-Positionen aktuell: u.a. Evotec, FMC, Gerresheimer, Hypoport, Nagarro, Nordex, Suss MicroTec | Wer Aktien leerverkauft, also auf fallende Kurse spekuliert (Shortselling), unterliegt bestimmten Transparenzpflichten. Aktuelle Meldungen über Leerverkäufe geben Aufschluss darüber. Diese Vorgaben... ► Artikel lesen | |
| BB BIOTECH | 48,100 | +0,42 % | Aktien KW 17 Märkte im Würgegriff. Planloser Trump , selbstbewusterer Iran. Lösungen müssen her. News. Rheinmetall. ITM Power. Porsche. The Platform Group. K+S. Bugatti. Circus. Surteco. NanoRepro. BB Biotech. Mensch und Maschine. ATOSS. MHP Hotel | Aktien - letzte Woche warteten die Märkte auf eine Lösung des Iran-Kriegs. Aber die Iraner sehen sich im Vorteil und Trump verlängert "auf unbestimmte Zeit" den Waffenstillstand. Aber die Strasse von... ► Artikel lesen | |
| MEDIGENE | 0,037 | +18,59 % | MEDIGENE AG im Fokus - ruhige Einordnung der Schwäche | ||
| QIAGEN | 30,290 | +0,65 % | QIAGEN N.V.: QIAGEN to advance AI-driven drug discovery with graph-based AI and curated bioinformatics knowledge with NVIDIA | Collaboration integrates NVIDIA accelerated computing and the NVIDIA BioNeMo platform with QIAGEN Digital Insights' curated biomedical knowledge bases Goal to advance how disease mechanisms... ► Artikel lesen | |
| MODERNA | 40,375 | -0,05 % | Moderna Flu Shot Heads To FDA Advisory Panel | ||
| PAION | 0,072 | -25,52 % | XFRA MISTRADE ANTRAG IN ISIN DE000A3E5EG5 WURDE STATTGEGEBEN | Dem Mistrade-Antrag wurde stattgegeben. Folgende Geschaefte in der ISIN DE000A3E5EG5, Wertpapier-Name: PAION AG INH O.N., wurden aufgehoben:ISIN Datum Zeit Volumen PreisDE000A3E5EG5 02.02.2026 08:13:40 300 0... ► Artikel lesen | |
| VALNEVA | 2,490 | +0,81 % | Valneva Aktie: Prognose kassiert - die Börse schaut woanders hin | Valneva hat das erste Quartal 2026 mit einem deutlichen Umsatzrückgang abgeschlossen und die Jahresprognose gesenkt. Der Aktienkurs reagierte nicht entscheidend, denn der Markt richtet seinen Blick... ► Artikel lesen | |
| AMGEN | 292,50 | +0,05 % | Endokrine Orbitopathie: Amgen lenkt den Blick auf Versorgungslücken und gesellschaftliche Folgen | München (ots) - Bis zu zwei Jahre Diagnose-Wartezeit und Beeinträchtigung von Lebensqualität, Beruf und sozialer Teilhabe[1-4]Zum Weltschilddrüsentag am 25. Mai 2026 rückt das Biotechnologieunternehmen... ► Artikel lesen | |
| EPIGENOMICS | 0,870 | 0,00 % | PTA-HV: Epigenomics AG: Einladung zur ordentlichen Hauptversammlung | DJ PTA-HV: Epigenomics AG: Einladung zur ordentlichen Hauptversammlung
Hauptversammlung gemäß -- 121 Abs. 4a AktG
Epigenomics AG: Einladung zur ordentlichen Hauptversammlung
Heidelberg... ► Artikel lesen | |
| NOVAVAX | 7,930 | 0,00 % | Novavax, Inc.: Novavax Reports First Quarter 2026 Financial Results and Operational Highlights | Total revenue of $140 million in the first quarter of 2026
Pfizer agreement announced in January for non-exclusive license to utilize Matrix-M® in two infectious... ► Artikel lesen | |
| STRYKER | 272,50 | -0,07 % | Stryker Buys Amplitude Vascular Systems For Upto $835 Mln To Expand Vascular Treatment Business | WASHINGTON (dpa-AFX) - Tuesday, BioStar Capital announced that Amplitude Vascular Systems, Inc., a medical technology company, has been acquired by Stryker Corporation (SYK) at a purchase price... ► Artikel lesen | |
| BIOGEN | 166,64 | -0,22 % | XETR DELETION OF INSTRUMENTS FROM XETRA - 22.05.2026 | The following instruments on XETRA do have their last trading day on 22.05.2026.Die folgenden Instrumente auf XETRA haben ihren letzten Handelstag am 22.05.2026..ISIN Name US09062X1037 Biogen Inc.US2521311074 DexCom... ► Artikel lesen | |
| BIOFRONTERA | 2,390 | -1,65 % | Biofrontera stock rating cut to Speculative Buy by Benchmark | ||
| HEIDELBERG PHARMA | 2,740 | -0,72 % | EQS-HV: Heidelberg Pharma AG: Bekanntmachung der Einberufung zur Hauptversammlung am 23.06.2026 in Ladenburg mit dem Ziel der europaweiten Verbreitung gemäß §121 AktG | EQS-News: Heidelberg Pharma AG
/ Bekanntmachung der Einberufung zur Hauptversammlung
Heidelberg Pharma AG: Bekanntmachung der Einberufung zur Hauptversammlung am 23.06.2026 in... ► Artikel lesen |
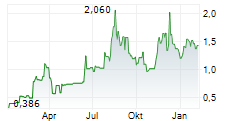
Das Unternehmen ASCLETIS PHARMA INC kann der Branche Biotechnologie zugeordnet werden. Mit einer aktuellen Kursveränderung von -0,040 (-2,33 %) liegt der Kurs bei 1,680 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu ASCLETIS PHARMA INC finden Sie auf Wallstreet Online.