| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
| 1,140 | 1,200 | 29.05. | |
| 1,130 | 1,200 | 29.05. |
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| Fr | CMS (00867): NEXT DAY DISCLOSURE RETURN | 2 | HKEx | ||
| Di | CMS (00867): VOLUNTARY AND BUSINESS UPDATE ANNOUNCEMENT APPROVAL OF DRUG CLINICAL TRIALS FOR ULCERATIVE COLITIS AND CROHN'S DISEASE OF INNOVATIVE DRUG ... | 1 | HKEx | ||
| 27.04. | Dividendenbekanntmachungen (27.04.2026) | 13.039 | wallstreetONLINE (Dividenden) | Unternehmen ISIN-Code Dividende (Währung) Dividende (EUR) ABN AMRO BANK NV ADR US00080Q1058 0,356 USD 0,3038 EUR AEON THANA SINSAP THAILAND PCL NVDR TH0664010R11 2,95 THB 0,0778 EUR AKZO... ► Artikel lesen | |
| 27.04. | China Medical System Holdings Limited: CMS (867.HK/8A8.SG) Signed An Exclusive Commercialization and Supply Agreement for Marketed Originator Intravenous Iron Products Monofer(R) and Cosmofer(R) | 394 | JCN Newswire | SHENZHEN, Apr 27, 2026 - (ACN Newswire) - China Medical System Holdings Limited ("CMS" or the "Group") is pleased to announce that, the Group through its wholly-owned subsidiary entered into an exclusive... ► Artikel lesen | |
| 27.04. | China Medical System Holdings Ltd.: CMS (867.HK/8A8.SG) Signed An Exclusive Commercialization and Supply Agreement for Marketed Originator Intravenous Iron Products Monofer and Cosmofer | 543 | GlobeNewswire (Europe) | SHENZHEN, CHINA, April 27, 2026 (GLOBE NEWSWIRE) -- China Medical System Holdings Limited ("CMS" or the "Group") is pleased to announce that, the Group through its wholly-owned subsidiary entered... ► Artikel lesen | |
| 23.04. | China Medical System Holdings Limited: CMS (867.HK; 8A8.SG): NDA for the Seasonal Allergic Rhinitis Indication of Class 1 Innovative Drug MG-K10 Accepted in China | 544 | JCN Newswire | SHENZHEN, Apr 23, 2026 - (ACN Newswire) - China Medical System Holdings Limited (the "Group" or "CMS") is pleased to announce that the New Drug Application (NDA) in China for the Seasonal Allergic Rhinitis... ► Artikel lesen | |
| 23.04. | CMS (00867): ANNOUNCEMENT ON VOTING RESULTS OF ANNUAL GENERAL MEETING HELD ON 23 APRIL 2026 | - | HKEx | ||
| 23.04. | CMS (00867): VOLUNTARY AND BUSINESS UPDATE ANNOUNCEMENT NEW DRUG APPLICATION FOR THE SEASONAL ALLERGIC RHINITIS INDICATION OF CLASS 1 INNOVATIVE DRUG ... | 1 | HKEx | ||
| CHINA MEDICAL SYSTEM Aktie jetzt für 0€ handeln | |||||
| 01.04. | CMS (00867): FORM OF PROXY | - | HKEx | ||
| 01.04. | CMS (00867): NOTICE OF ANNUAL GENERAL MEETING | - | HKEx | ||
| 01.04. | CMS (00867): PROPOSALS FOR (1) RE-ELECTION OF THE RETIRING DIRECTORS (2) DECLARATION OF FINAL DIVIDEND (3) GENERAL MANDATES TO ISSUE SHARES AND TO REPURCHASE ... | - | HKEx | ||
| 01.04. | CMS (00867): ENVIRONMENTAL, SOCIAL AND GOVERNANCE REPORT 2025 | - | HKEx | ||
| 01.04. | CMS (00867): ANNUAL REPORT 2025 | - | HKEx | ||
| 17.03. | CMS (00867): TERMS OF REFERENCE OF THE NOMINATION COMMITTEE | 2 | HKEx | ||
| 17.03. | CMS (00867): LIST OF DIRECTORS AND THEIR ROLE AND FUNCTION | 1 | HKEx | ||
| 17.03. | CMS (00867): FINAL DIVIDEND FOR THE YEAR ENDED 31 DECEMBER 2025 | 1 | HKEx | ||
| 16.03. | China Medical System Holdings Limited Full Year Income Falls | 1 | RTTNews | ||
| 16.03. | CMS (00867): ANNUAL RESULTS ANNOUNCEMENT FOR THE YEAR ENDED 31 DECEMBER 2025 AND CHANGE OF MEMBER OF THE ENVIRONMENTAL, SOCIAL AND GOVERNANCE COMMITTEE | 1 | HKEx | ||
| 13.03. | China Medical System Holdings Ltd.: CMS(867.HK/8A8.SG): New Drug for Renal Anaemia Desidustat Tablets Approved in China | 2 | GlobeNewswire (USA) | ||
| 13.03. | CMS (00867): VOLUNTARY AND BUSINESS UPDATE ANNOUNCEMENT NEW DRUG FOR RENAL ANAEMIA DESIDUSTAT TABLETS APPROVED IN CHINA | 1 | HKEx |
1 2 3 Weiter >>
42 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| FRESENIUS | 36,270 | -1,20 % | Dividendenbekanntmachungen (25.05.2026) | Unternehmen ISIN-Code Dividende (Währung) Dividende (EUR) AQUILA SA FR0010340711 - 0,25 EUR ARISTOCRAT LEISURE LIMITED AU000000ALL7 0,5 AUD 0,3071 EUR ARKEMA SA FR0010313833 - 3,6 EUR BLEECKER... ► Artikel lesen | |
| FRESENIUS MEDICAL CARE | 37,190 | -0,13 % | FMC startet Aktienrückkauf über eine Milliarde Euro | Die Fresenius Medical Care AG hat ein Aktienrückkaufprogramm mit einem Gesamtvolumen von rund einer Milliarde Euro beschlossen, das innerhalb von zwölf Monaten in Tranchen umgesetzt werden soll. Grundlage... ► Artikel lesen | |
| SIEMENS HEALTHINEERS | 34,920 | +0,11 % | EQS-CMS: Siemens Healthineers AG: Veröffentlichung einer Kapitalmarktinformation | EQS Zulassungsfolgepflichtmitteilung: Siemens Healthineers AG
/ Bekanntmachung nach Art. 5 Abs. 1 lit. a) der Verordnung (EU) Nr. 596/2014, zuletzt geändert durch die Verordnung (EU)... ► Artikel lesen | |
| CARL ZEISS MEDITEC | 25,880 | -2,19 % | EQS-DD: Carl Zeiss Meditec AG: Peter Kameritsch, Kauf | Meldung und öffentliche Bekanntgabe der Geschäfte von Personen, die Führungsaufgaben wahrnehmen, sowie in enger Beziehung zu ihnen stehenden Personen
28.05.2026 / 14:57... ► Artikel lesen | |
| ECKERT & ZIEGLER | 16,520 | +1,98 % | EQS-News: Eckert & Ziegler SE: Eckert & Ziegler geht Partnerschaft mit Thor Medical ein, um der pharmazeutischen Industrie den Zugang zu Blei-212 zu ermöglichen | EQS-News: Eckert & Ziegler SE
/ Schlagwort(e): Vereinbarung/Kooperation
Eckert & Ziegler geht Partnerschaft mit Thor Medical ein, um der pharmazeutischen Industrie den Zugang... ► Artikel lesen | |
| UNITEDHEALTH | 324,60 | -0,43 % | Massachusetts AG sues UnitedHealthcare over alleged Medicaid fraud | ||
| GERRESHEIMER | 27,200 | -2,51 % | PRESSESPIEGEL/Unternehmen: INFINEON, SIEMENS, KNDS, GERRESHEIMER, MANGO | DJ PRESSESPIEGEL/Unternehmen
Die wirtschaftsrelevanten Themen aus den Medien, zusammengestellt von Dow Jones Newswires.
INFINEON - Die Zeit der milliardenteuren Neubauten bei Infineon endet.... ► Artikel lesen | |
| DRAEGERWERK | 91,80 | -1,08 % | Drägerwerk-Aktie im Fokus: Wochengewinner im SDAX in KW 21 | ||
| INTUITIVE SURGICAL | 363,90 | +0,01 % | Intuitive Surgical, Inc.: Intuitive Announces New Chief Commercial and Marketing Officer Taylor Patton | SUNNYVALE, Calif., May 28, 2026 (GLOBE NEWSWIRE) -- Intuitive (NASDAQ: ISRG), a global technology leader in minimally invasive care and the pioneer of robotic-assisted surgery, today announced the... ► Artikel lesen | |
| MEDTRONIC | 63,24 | -0,06 % | Medtronic baut europäischen Software-Hub für vernetzte Herzgeräte | ||
| THERMO FISHER | 422,70 | +0,09 % | Laufende Fusionskontrollverfahren: Astorg Group S.à.r.l., Luxemburg; mittelbarer Erwerb alleiniger Kontrolle über den Mikrobiologie-Geschäftsbereich von Thermo Fisher Scientific Inc., Waltham/MA/USA (u.a. Oxoid Deutschland GmbH) | Datum der Anmeldung:22.05.2026Aktenzeichen:B3-68/26Unternehmen:Astorg Group S.à.r.l., Luxemburg; mittelbarer Erwerb alleiniger Kontrolle über den Mikrobiologie-Geschäftsbereich von Thermo Fisher Scientific... ► Artikel lesen | |
| TELADOC HEALTH | 6,600 | +1,07 % | TDOC Stock Pops as Teladoc Teams Up With Walmart | ||
| STRATEC | 17,680 | +1,96 % | Stratec-Aktie: Hier zählt die Zukunft | Die Aktie von Stratec befindet sich weiterhin in einer schwierigen Marktphase. Seit dem Hoch im Oktober bei rund 30 € dominiert ein langfristiger Abwärtstrend, der lediglich von zwischenzeitlichen Erholungsbewegungen... ► Artikel lesen | |
| RHOEN-KLINIKUM | 12,300 | -1,60 % | Dividendenbekanntmachungen (28.05.2026) | Unternehmen ISIN-Code Dividende (Währung) Dividende (EUR) ACTIVIA PROPERTIES INC JP3047490002 3169 JPY 17,0809 EUR ADVANCED MEDICAL SOLUTIONS GROUP PLC GB0004536594 0,0201 GBP 0,0232 EUR AIA... ► Artikel lesen | |
| GENEDX | 51,99 | 0,00 % | GeneDx (WGS) Fell Despite Continued Execution Improvement |
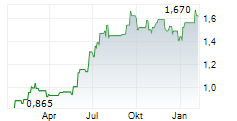
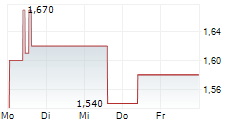
Das Unternehmen CHINA MEDICAL SYSTEM HOLDINGS LTD kann der Branche Gesundheitswesen zugeordnet werden. Die relative Kursveränderung beträgt aktuell -3,42 % bei einem Aktienkurs von 1,130 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu CHINA MEDICAL SYSTEM HOLDINGS LTD finden Sie auf Wallstreet Online.