| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
| 0,454 | 0,495 | 19:08 | |
| 0,453 | 0,494 | 19:07 |
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| Mi | CSTONE PHARMA-B (02616): VOLUNTARY ANNOUNCEMENT - JOINT SHARE PURCHASE PLAN INITIATED BY THE DIRECTORS AND SENIOR MANAGEMENT | 1 | HKEx | ||
| 01.06. | CSTONE PHARMA-B (02616): FORM OF PROXY FOR ANNUAL GENERAL MEETING | 3 | HKEx | ||
| CSTONE PHARMACEUTICALS Aktie jetzt für 0€ handeln | |||||
| 01.06. | CSTONE PHARMA-B (02616): NOTICE OF ANNUAL GENERAL MEETING | 3 | HKEx | ||
| 01.06. | CSTONE PHARMA-B (02616): PROPOSALS FOR GENERAL MANDATES TO ISSUE AND BUY BACK SHARES, RE-ELECTION OF RETIRING DIRECTORS, RE-APPOINTMENT OF AUDITOR, PROPOSED ... | 4 | HKEx | ||
| 01.06. | CSTONE PHARMA-B Plans to Launch First Global Multi-center Phase III Trial of CS2009 by End-2026 | 8 | AASTOCKS | ||
| 01.06. | CSTONE PHARMA-B (02616): VOLUNTARY ANNOUNCEMENT - ASCO 2026 | CSTONE PRESENTS LATEST CS2009 (PD-1/VEGF/CTLA-4 TRISPECIFIC ANTIBODY) CLINICAL DATA | 6 | HKEx | ||
| 14.05. | CSTONE PHARMA-B Plans to Repurchase Up to 136 Million Shares on Open Market | 8 | AASTOCKS | ||
| 13.05. | CSTONE PHARMA-B (02616): VOLUNTARY ANNOUNCEMENT - ON-MARKET SHARE REPURCHASE | 2 | HKEx | ||
| 11.05. | CSTONE PHARMA-B Launches Nationwide Commercial Shipment of Locally Manufactured Pralsetinib Capsules | 3 | AASTOCKS | ||
| 11.05. | CSTONE PHARMA-B (02616): VOLUNTARY ANNOUNCEMENT - CSTONE INITIATES NATIONWIDE COMMERCIAL SUPPLY OF LOCALLY MANUFACTURED PRALSETINIB CAPSULES | 6 | HKEx | ||
| 27.04. | CSTONE PHARMA-B (02616): ENVIRONMENTAL, SOCIAL AND GOVERNANCE REPORT 2025 | 3 | HKEx | ||
| 27.04. | CSTONE PHARMA-B (02616): 2025 ANNUAL REPORT | - | HKEx | ||
| 22.04. | CSTONE PHARMA-B (02616): COMPLETION OF PLACING OF NEW SHARES UNDER GENERAL MANDATE | 1 | HKEx | ||
| 22.04. | CSTONE PHARMA-B (02616): NEXT DAY DISCLOSURE RETURN | 1 | HKEx | ||
| 22.04. | CSTONE PHARMA-B (02616): VOLUNTARY ANNOUNCEMENT - ASCO 2026 PREVIEW: CSTONE TO PRESENT UPDATED CLINICAL DATA FOR CS2009 (PD 1/VEGF/CTLA 4 TRISPECIFIC ... | 3 | HKEx | ||
| 20.04. | CStone stellt neue ADC-Daten auf AACR-Jahrestagung vor: Aktie im Höhenflug | 7 | Investing.com Deutsch | ||
| 20.04. | CStone presents preclinical data on three ADC candidates at AACR | 1 | Investing.com | ||
| 20.04. | CSTONE PHARMA-B (02616): VOLUNTARY ANNOUNCEMENT - CSTONE PRESENTED PRECLINICAL DATA FOR THREE NOVEL OR DIFFERENTIATED ADCS AT AACR 2026, INCLUDING CS5007 ... | 1 | HKEx | ||
| 15.04. | CSTONE PHARMA-B (02616): PLACING OF NEW SHARES UNDER GENERAL MANDATE | 2 | HKEx | ||
| 02.04. | CSTONE PHARMA-B (02616): LETTER AND REPLY FORM TO REGISTERED SHAREHOLDERS - ARRANGEMENT OF ELECTRONIC DISSEMINATION OF CORPORATE COMMUNICATIONS | 4 | HKEx |
1 2 Weiter >>
35 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BIONTECH | 73,95 | -0,27 % | Neue CAR-T-Erfolge bei Autoimmunerkrankungen: Warum Lösungen für Zelltherapien vor dem nächsten Wachstumsschub stehen könnten | ||
| EVOTEC | 4,750 | 0,00 % | EQS-News: Evotec SE: Evotec ernennt Rui Wang zum Head of Global In Silico & AI | EQS-News: Evotec SE
/ Schlagwort(e): Sonstiges
Evotec ernennt Rui Wang zum Head of Global In Silico & AI
02.06.2026 / 07:30 CET/CEST
Für den Inhalt der Mitteilung ist... ► Artikel lesen | |
| BB BIOTECH | 44,950 | -0,77 % | Aktien KW 17 Märkte im Würgegriff. Planloser Trump , selbstbewusterer Iran. Lösungen müssen her. News. Rheinmetall. ITM Power. Porsche. The Platform Group. K+S. Bugatti. Circus. Surteco. NanoRepro. BB Biotech. Mensch und Maschine. ATOSS. MHP Hotel | Aktien - letzte Woche warteten die Märkte auf eine Lösung des Iran-Kriegs. Aber die Iraner sehen sich im Vorteil und Trump verlängert "auf unbestimmte Zeit" den Waffenstillstand. Aber die Strasse von... ► Artikel lesen | |
| MEDIGENE | 0,021 | 0,00 % | PTA-Adhoc: Medigene AG: Widerruf der Zulassung der Aktien der Medigene AG zum Handel im regulierten Markt | DJ PTA-Adhoc: Medigene AG: Widerruf der Zulassung der Aktien der Medigene AG zum Handel im regulierten Markt
Veröffentlichung von Insiderinformationen gemäß Artikel 17 MAR Schlagwort(e): Sonstiges... ► Artikel lesen | |
| QIAGEN | 31,740 | -0,02 % | DEUTSCHE BANK RESEARCH stuft QIAGEN NV auf 'Buy' | FRANKFURT (dpa-AFX Analyser) - Deutsche Bank Research hat Qiagen mit einem Kursziel von 43 US-Dollar auf "Buy" belassen. Das Feedback aus Gesprächen mit etlichen US-Laborexperten deute darauf hin, dass... ► Artikel lesen | |
| MODERNA | 39,765 | -3,68 % | Moderna jab on trial for cancer-causing syndrome | ||
| PAION | 0,062 | -23,60 % | XFRA MISTRADE ANTRAG IN ISIN DE000A3E5EG5 WURDE STATTGEGEBEN | Dem Mistrade-Antrag wurde stattgegeben. Folgende Geschaefte in der ISIN DE000A3E5EG5, Wertpapier-Name: PAION AG INH O.N., wurden aufgehoben:ISIN Datum Zeit Volumen PreisDE000A3E5EG5 02.02.2026 08:13:40 300 0... ► Artikel lesen | |
| VALNEVA | 2,265 | -2,33 % | EILMELDUNG bei Valneva: Der Kurs nähert sich einer gefährlichen Marke - So reagieren erfahrene Anleger jetzt | ||
| AMGEN | 298,20 | -0,52 % | EQS-News: Amgen: Europäische Kommission erteilt Zulassung für Imdylltra von Amgen für die Behandlung von kleinzelligem Lungenkrebs im fortgeschrittenen Stadium | EQS-News: Amgen
/ Schlagwort(e): Sonstiges/Sonstiges
EUROPÄISCHE KOMMISSION ERTEILT ZULASSUNG FÜR IMDYLLTRA VON AMGEN FÜR DIE BEHANDLUNG VON KLEINZELLIGEM... ► Artikel lesen | |
| NOVAVAX | 7,300 | -2,14 % | NOVAVAX INC: Stabilität vor der nächsten Bewegung | ||
| STRYKER | 270,90 | +3,56 % | Leerink cuts Stryker stock price target to $407 on model update | ||
| BIOGEN | 169,08 | +1,18 % | Biogen's Salanersen Wins FDA Breakthrough Therapy Designation For Spinal Muscular Atrophy Treatment | WESTON (dpa-AFX) - Thursday, Biogen Inc. (BIIB) announced that the U.S. Food and Drug Administration has granted salanersen Breakthrough Therapy Designation for the treatment of spinal muscular... ► Artikel lesen | |
| BIOFRONTERA | 2,600 | +1,56 % | BIOFRONTERA AG zeigt bemerkenswerte Stärke | ||
| HEIDELBERG PHARMA | 2,710 | +4,23 % | HEIDELBERG PHARMA AG: Ruhe vor der nächsten Phase | ||
| ILLUMINA | 135,24 | -1,36 % | Illumina launches StrataMap Spatial research solution |
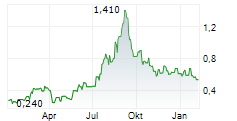
Das Unternehmen CSTONE PHARMACEUTICALS CO LTD kann der Branche Biotechnologie zugeordnet werden. Die relative Kursveränderung beträgt aktuell -2,33 % bei einem Aktienkurs von 0,470 Euro. Beim derzeitigen Kurs ergibt sich, basierend auf einer gesamten Ausschüttung von 0,00 Euro in den vergangenen 12 Monaten, eine Dividendendrendite von 0,00 %.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu CSTONE PHARMACEUTICALS CO LTD finden Sie auf Wallstreet Online.