| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
| 3,097 | 3,217 | 10:29 | |
| 3,097 | 3,217 | 10:29 |
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| Fr | CMSI: Three Reasons for Recent Underperformance of Pharma Sector vs HSI; Top Picks INNOVENT BIO , HANSOH PHARMA and KEYMED BIO-B | 7 | AASTOCKS | ||
| Do | HANSOH PHARMA (03692): 2026 SHARE SCHEME | 3 | HKEx | ||
| HANSOH PHARMACEUTICAL Aktie jetzt für 0€ handeln | |||||
| Do | HANSOH PHARMA (03692): NOTIFICATION LETTER TO NON-REGISTERED HOLDERS AND REQUEST FORM | 2 | HKEx | ||
| Do | HANSOH PHARMA (03692): NOTIFICATION LETTER TO REGISTERED SHAREHOLDERS AND REPLY FORM | 3 | HKEx | ||
| Do | HANSOH PHARMA (03692): FORM OF PROXY FOR THE ANNUAL GENERAL MEETING TO BE HELD ON FRIDAY, JUNE 26, 2026 | 2 | HKEx | ||
| Do | HANSOH PHARMA (03692): NOTICE OF ANNUAL GENERAL MEETING | 1 | HKEx | ||
| Do | HANSOH PHARMA (03692): (1) PROPOSED RE-ELECTION OF DIRECTORS, (2) PROPOSED GRANTING OF GENERAL MANDATES TO REPURCHASE SHARES AND TO ISSUE SHARES, (3) ... | 3 | HKEx | ||
| Do | HANSOH PHARMA 's Olipudase Injection NDA Accepted by NMPA in China | 3 | AASTOCKS | ||
| Do | HANSOH PHARMA (03692): VOLUNTARY ANNOUNCEMENT - NEW DRUG APPLICATION OF OLATOREPATIDE INJECTION WAS ACCEPTED BY THE NATIONAL MEDICAL PRODUCTS ADMINISTRATION | 3 | HKEx | ||
| 22.05. | HSCEI Adds HANSOH PHARMA and AKESO, Removes SUNNY OPTICAL and HAIER SMARTHOME in Quarterly Review | 10 | AASTOCKS | ||
| 22.05. | HANSOH PHARMA Drug for Non-Small Cell Lung Cancer Included as Breakthrough Therapy | 4 | AASTOCKS | ||
| 21.05. | HANSOH PHARMA (03692): VOLUNTARY ANNOUNCEMENT - THE NMPA HAS GRANTED BREAKTHROUGH-THERAPY-DESIGNATED DRUG FOR FOURTH-GENERATION EGFR-TKI HS-10504 TABLETS ... | 3 | HKEx | ||
| 15.05. | HANSOH PHARMA (03692): CHANGES OF JOINT COMPANY SECRETARY, AUTHORIZED REPRESENTATIVE AND PROCESS AGENT | 1 | HKEx | ||
| 14.05. | HANSOH PHARMA HS-10541 Tablet Approved for Clinical Trial | 3 | AASTOCKS | ||
| 13.05. | HANSOH PHARMA (03692): VOLUNTARY ANNOUNCEMENT - THE CLINICAL TRIAL APPROVAL FOR HS-10541 TABLETS ISSUED BY THE NATIONAL MEDICAL PRODUCTS ADMINISTRATION | 2 | HKEx | ||
| 11.05. | HANSOH PHARMA HS-20093 for Esophageal Squamous Cell Carcinoma Included as Breakthrough Therapy Drug | 2 | AASTOCKS | ||
| 11.05. | HANSOH PHARMA (03692): VOLUNTARY ANNOUNCEMENT - THE NMPA HAS GRANTED BREAKTHROUGH-THERAPY-DESIGNATED DRUG FOR B7-H3-TARGETED ANTIBODY-DRUG CONJUGATE HS-20093 ... | 2 | HKEx | ||
| 30.04. | HANSOH PHARMA (03692): VOLUNTARY ANNOUNCEMENT - SHARE PURCHASE PURSUANT TO THE RESTRICTED SHARE UNIT SCHEME | 2 | HKEx | ||
| 29.04. | HANSOH PHARMA (03692): VOLUNTARY ANNOUNCEMENT - THE NMPA HAS GRANTED BREAKTHROUGH-THERAPY-DESIGNATED DRUG FOR B7-H3-TARGETED ANTIBODY-DRUG CONJUGATE HS-20093 ... | 2 | HKEx | ||
| 29.04. | HANSOH PHARMA (03692): VOLUNTARY ANNOUNCEMENT - THE CLINICAL TRIAL APPROVALS FOR HS-10522 TABLETS ISSUED BY THE NATIONAL MEDICAL PRODUCTS ADMINISTRATION | 2 | HKEx |
1 2 3 Weiter >>
49 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BAYER | 35,210 | -1,34 % | Bayer-Aktie stürzt 5% ab: Schwindet die Hoffnung auf eine Glyphosat-Einigung komplett? | Der Leverkusener Chemie- und Pharmakonzern Bayer erleidet an der Börse einen herben Rückschlag. Nach einer Phase der zwischenzeitlichen Erholung ist das Papier des DAX-Unternehmens im jüngsten Frankfurter... ► Artikel lesen | |
| NOVO NORDISK | 36,025 | +0,92 % | Börsenrotation ohne KI! Jetzt 100 % mit MustGrow, Bayer und Novo Nordisk, Achtung bei K+S | Erstellt und veröffentlicht im Auftrag von MustGrow Biologics Corp. Perspektivenwechsel bleibt das Gebot der Stunde. Denn gestern tagte die EZB zur aktuellen Lage bei Inflation und Zinsniveau. Mit einer... ► Artikel lesen | |
| PFIZER | 22,225 | +0,04 % | Rendite-Chance Krebsmarkt: Mit BioNTech, Vidac Pharma und Pfizer den nächsten Mrd.-Trigger erwischen | Die Alterung der Gesellschaft lässt die Gesundheitsausgaben rasant steigen. Im Krebssegment zeichnet sich ein Billionenmarkt ab, denn bis 2040 werden jährlich über 30 Mio. Neuerkrankungen erwartet.... ► Artikel lesen | |
| NOVARTIS | 126,96 | +0,19 % | Neue CAR-T-Erfolge bei Autoimmunerkrankungen: Warum Lösungen für Zelltherapien vor dem nächsten Wachstumsschub stehen könnten | ||
| MERCK KGAA | 138,10 | +0,47 % | EQS-PVR: Merck KGaA: Veröffentlichung gemäß § 40 Abs. 1 WpHG mit dem Ziel der europaweiten Verbreitung | EQS Stimmrechtsmitteilung: Merck KGaA
Merck KGaA: Veröffentlichung gemäß § 40 Abs. 1 WpHG mit dem Ziel der europaweiten Verbreitung
08.06.2026 / 16:57 CET/CEST
Veröffentlichung... ► Artikel lesen | |
| GILEAD SCIENCES | 111,02 | -0,09 % | Merck, Gilead share positive phase 3 results from combination HIV pill | ||
| AURORA CANNABIS | 2,920 | -1,42 % | Aurora Deepens its Impact for Veterans Across Canada | ||
| SANOFI | 76,96 | +1,16 % | JEFFERIES stuft SANOFI auf 'Buy' | NEW YORK (dpa-AFX Analyser) - Das Analysehaus Jefferies hat die Einstufung für Sanofi mit einem Kursziel von 100 Euro auf "Buy" belassen. Bei den Franzosen gebe es trotz des Führungswechsels keinen... ► Artikel lesen | |
| GSK | 21,565 | -2,42 % | DEUTSCHE BANK RESEARCH stuft GSK auf 'Hold' | FRANKFURT (dpa-AFX Analyser) - Deutsche Bank Research hat die Einstufung für GSK mit einem Kursziel von 1900 Pence auf "Hold" belassen. Emmanuel Papadakis bezeichnete in seinem am Dienstag vorliegenden... ► Artikel lesen | |
| ABBVIE | 192,50 | -0,47 % | Drei Qualitätswerte im Fokus: Was der "Quant Growth And Income"-Ansatz jetzt über Microsoft, Visa und AbbVie verrät | Microsoft, Visa und AbbVie stehen im Zentrum eines quantitativen Wachstums- und Einkommensansatzes, den Seeking Alpha aktuell transparent macht. Der Einblick in das "Quant Growth And Income"-Portfolio... ► Artikel lesen | |
| ROCHE | 345,51 | -1,22 % | JEFFERIES stuft ROCHE HOLDINGS AG auf 'Underperform' | NEW YORK (dpa-AFX Analyser) - Das Analysehaus Jefferies hat die Einstufung für Roche mit einem Kursziel von 230 Franken auf "Underperform" belassen. Das Risiko für die mittelfristigen Ziele der Schweizer... ► Artikel lesen | |
| CANOPY GROWTH | 0,892 | +0,38 % | Canopy Growth Just Made a Big Acquisition: Game-Changer -- or Another Misstep? | ||
| ELI LILLY | 1.004,20 | +0,80 % | Eli Lilly: Pharma-Gigant greift nach der Billionen-Dollar-Krone! | Zwischen astronomischem Wachstum und charttechnischem Momentum! Rückblick Die Story von Eli Lilly lässt sich im Moment in drei Buchstaben zusammenfassen: GLP-1. Das Unternehmen liefert sich ein spektakuläres... ► Artikel lesen | |
| MERCK & CO | 103,30 | -0,33 % | Gilead and Merck's once-weekly HIV pill meets trial goals | ||
| ASTRAZENECA | 156,90 | -0,35 % | Evotec holt KI-Manager von AstraZeneca | Evotec verstärkt den Bereich Künstliche Intelligenz und computergestützte Forschung mit einem erfahrenen Branchenmanager. Rui Wang übernimmt die neu geschaffene Position des Head of Global In Silico... ► Artikel lesen |
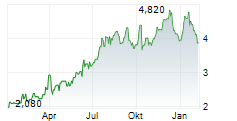
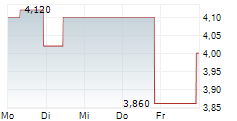
Das Unternehmen HANSOH PHARMACEUTICAL GROUP CO LTD kann der Branche Pharma zugeordnet werden. Mit einer aktuellen Kursveränderung von +0,048 (+1,51 %) liegt der Kurs bei 3,219 Euro. Beim derzeitigen Kurs ergibt sich, basierend auf einer gesamten Ausschüttung von 0,04 Euro in den vergangenen 12 Monaten, eine Dividendendrendite von +1,24 %.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu HANSOH PHARMACEUTICAL GROUP CO LTD finden Sie auf Wallstreet Online.