- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 30.04. | Lyra Therapeutics, Inc. - 15-12G, Securities registration termination | 9 | SEC Filings | ||
| LYRA THERAPEUTICS Aktie jetzt für 0€ handeln | |||||
| 20.04. | Lyra Therapeutics, Inc. - 25, Notification of the removal from listing and registration of matured, redeemed or retired securities | - | SEC Filings | ||
| 17.04. | Lyra Therapeutics to delist from Nasdaq, suspend SEC reporting | 1 | Investing.com | ||
| 17.04. | Lyra Therapeutics, Inc. Announces Intention to Voluntarily File Form 25 for Delisting from Nasdaq and Subsequent Deregistration with the SEC | 191 | GlobeNewswire (Europe) | WATERTOWN, Mass., April 17, 2026 (GLOBE NEWSWIRE) -- Lyra Therapeutics, Inc. (the "Company") today announced its intention to voluntarily file a Form 25 Notification of Delisting with the U.S. Securities... ► Artikel lesen | |
| 17.04. | Lyra Therapeutics, Inc. - 8-K, Current Report | - | SEC Filings | ||
| 10.04. | Lyra Therapeutics, Inc. - 8-K, Current Report | - | SEC Filings | ||
| 31.03. | Lyra Therapeutics, Inc. - 10-K, Annual Report | - | SEC Filings | ||
| 16.03. | Lyra Therapeutics, Inc. - 8-K, Current Report | 1 | SEC Filings | ||
| 27.02. | Lyra Therapeutics, Inc. - 8-K, Current Report | 1 | SEC Filings | ||
| 06.02. | Lyra Therapeutics, Inc. - 8-K, Current Report | 1 | SEC Filings | ||
| 16.01. | Weekly Buzz: Lyra Therapeutics Slashes Jobs; FDA Okays Fortress Biotech's ZYCUBO; Boston Scientific To Acquire Penumbra; BriaCell Therapeutics On Watch | 821 | AFX News | NEW BRUNSWICK (dpa-AFX) - This week, the biotech sector saw a steady stream of high-impact news, including a major workforce reduction and a fresh round of regulatory clearances spanning diabetes... ► Artikel lesen | |
| 12.01. | Gold Rises Sharply; Lyra Therapeutics Shares Plunge | 1 | Benzinga.com | ||
| 12.01. | Lyra Therapeutics, Inc. - 8-K, Current Report | - | SEC Filings | ||
| 12.11.25 | Lyra Therapeutics Reports Third Quarter 2025 Financial Results and Provides Corporate Update | 345 | GlobeNewswire (Europe) | - Company plans new, confirmatory Phase 3 clinical trial of LYR-210 for treatment of chronic rhinosinusitis (CRS) without nasal polyps, on path to NDA submission - WATERTOWN, Mass., Nov. 12, 2025... ► Artikel lesen | |
| 06.10.25 | Lyra Therapeutics Announces Clinical Plan for LYR-210 and Late-Breaking Oral Presentation of Phase 3 ENLIGHTEN 2 Study | 417 | GlobeNewswire (Europe) | Company outlines clinical plan for LYR-210 based on FDA meeting on the path to NDA submission Positive ENLIGHTEN 2 results highlighted at the AAO-HNS 2025 Annual Meeting WATERTOWN, Mass., Oct. 06... ► Artikel lesen | |
| 12.08.25 | Lyra Therapeutics Reports Second Quarter 2025 Financial Results and Provides Corporate Update | 288 | GlobeNewswire (Europe) | - Positive ENLIGHTEN 2 Phase 3 results announced in June 2025 propel business activities in manufacturing, regulatory strategy and ENLIGHTEN clinical dataset analysis to advance path forward for LYR-210... ► Artikel lesen | |
| 02.06.25 | Lyra Therapeutics Reports Positive Results from the ENLIGHTEN 2 Phase 3 Trial of LYR-210 Achieving Statistically Significant Results for Primary and Key Secondary Endpoints in the Treatment of Chronic Rhinosinusitis (CRS) | 393 | GlobeNewswire (Europe) | ENLIGHTEN 2 trial met primary endpoint with LYR-210 demonstrating statistically significant improvement in the composite of the three cardinal symptoms (3CS) of CRS at 24 weeks (p=0.0078)ENLIGHTEN... ► Artikel lesen |
17 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BAYER | 37,980 | -1,12 % | Dax vor turbulentem Wochenausklang: Bayer - Das wird jetzt für die Aktie entscheidend | Dem Dax steht einmal mehr ein turbulenter Wochenausklang bevor. Bislang gestaltete sich die Handelswoche positiv. Ein robuster Wochenausklang würde das zuletzt zu beobachtende Erholungsszenario bestätigen... ► Artikel lesen | |
| NOVO NORDISK | 38,390 | -0,90 % | Neue Studiendaten sorgen für Fantasie im Milliardenmarkt der Lebermedizin: Novo Nordisk-Aktie im Fokus: Semaglutid könnte den nächsten Milliardenmarkt erobern | Novo Nordisk hat neue Studiendaten zu Semaglutid veröffentlicht und damit erneut Aufmerksamkeit an den Finanzmärkten ausgelöst. Der Wirkstoff, der bereits den Boom bei Adipositas- und Diabetesmedikamenten... ► Artikel lesen | |
| PFIZER | 22,295 | -0,60 % | Unterbewertet - Analysten rücken LifeScience in den Fokus! Bayer, Vidac Pharma, Novo Nordisk und Pfizer im Check | Dass Analysten den LifeScience-Bereich zuletzt wieder besser einschätzen, zeigt die dynamische Kursziel-Anpassung der DZ Bank für Bayer. Der starke Jahresstart des Pharma- und Agrarchemiekonzerns führt... ► Artikel lesen | |
| NOVARTIS | 130,60 | -2,07 % | Aktien Europa Schluss: Knappe Gewinne - SMI in der Schweiz legt stärker zu | PARIS/LONDON/ZÜRICH (dpa-AFX) - Europas wichtigste Aktienmärkte sind am Dienstag mehrheitlich wenig von der Stelle gekommen. Angesichts schwächelnder US-Börsen behauptete der Eurozonen-Leitindex EuroStoxx... ► Artikel lesen | |
| MERCK KGAA | 127,50 | -1,92 % | JEFFERIES stuft MERCK KGAA auf 'Hold' | NEW YORK (dpa-AFX Analyser) - Jefferies hat das Kursziel für Merck KGaA nach einem Analystenwechsel von 180 auf 129 Euro gesenkt und die Aktien von "Buy" auf "Hold" abgestuft. Das untere Ende der Zielspanne... ► Artikel lesen | |
| GILEAD SCIENCES | 115,50 | -0,48 % | Gilead Gets Positive CHMP Opinion For Trodelvy In First-Line Metastatic TNBC | FOSTER CITY (dpa-AFX) - Gilead Sciences Inc. (GILD) on Friday said the European Medicines Agency's Committee for Medicinal Products for Human Use recommended marketing authorization for Trodelvy... ► Artikel lesen | |
| SANOFI | 76,88 | -1,14 % | Sanofi Reports Positive Phase 2 Data For Efdoralprin Alfa In AATD-Related Emphysema | PARIS (dpa-AFX) - Sanofi SA (SNY) reported positive data from the Phase 2 ElevAATe study of efdoralprin alfa in adult patients with alpha-1 antitrypsin deficiency-related emphysema.Alpha-1 Antitrypsin... ► Artikel lesen | |
| ABBVIE | 186,20 | -0,19 % | AbbVie's MAVIRET Secures Positive CHMP Opinion For Acute Hepatitis C Infection Treatment | WASHINGTON (dpa-AFX) - Friday, AbbVie Inc. (ABBV) announced that the European Medicines Agency's Committee for Medicinal Products for Human Use has adopted a positive opinion recommending the... ► Artikel lesen | |
| STADA ARZNEIMITTEL | - | - | Enzymatica AB: Quarterly report Q1/2026 - Enzymatica strengthens international position through STADA agreement | First quarter
Net sales amounted to SEK 11.4 million (12.3).
Operating result amounted to SEK -13.6.million (-18.0)
Earnings per share, before... ► Artikel lesen | |
| ELI LILLY | 931,30 | +0,84 % | Eli Lilly vor dem nächsten Kursschub? Technische Analyse signalisiert begrenztes Abwärtsrisiko und neue Höchststände | Eli Lilly bleibt aus technischer Sicht ein attraktiver Large Cap mit intaktem Aufwärtstrend und begrenztem Abwärtsrisiko. Die jüngste Konsolidierung nach dem steilen Anstieg wird im Kern als normale... ► Artikel lesen | |
| MERCK & CO | 104,80 | -0,57 % | Ihre wichtigsten Termine: Alle Analysten-Augen auf: Aroundtown, Kingfisher sowie Merck & Co | © Foto: © 2023 Merck & CoGut geplant in den Tag. Mit dem wO-Tagesausblick haben Sie die wichtigsten Termine im Blick und starten bestens vorbereitet in den neuen Handelstag.Unternehmenstermine 06:45... ► Artikel lesen | |
| ASTRAZENECA | 161,90 | -1,28 % | DEUTSCHE BANK RESEARCH stuft ASTRAZENECA auf 'Sell' | FRANKFURT (dpa-AFX Analyser) - Deutsche Bank Research hat die Einstufung für Astrazeneca mit einem Kursziel von 11500 Pence auf "Sell" belassen. Die Erwartungen an die Phase-III-Studie Calypso mit dem... ► Artikel lesen | |
| BRISTOL-MYERS SQUIBB | 51,08 | -0,16 % | Bristol Myers Squibb Enters Agreement With Anthropic To Deploy Claude | NEW YORK CITY (dpa-AFX) - Bristol Myers Squibb (BMY) announced a strategic agreement with Anthropic to deploy Claude across the company's research, clinical development, manufacturing, commercial... ► Artikel lesen | |
| TEVA | 29,800 | -0,33 % | Teva: European Medicines Agency Accepts Marketing Authorization Application For TEV-'749 | PETAH TIKVA (dpa-AFX) - Teva Pharmaceuticals International GmbH, a unit of Teva Pharmaceutical Industries (TEVA), and Medincell announced that the European Medicines Agency has accepted the... ► Artikel lesen | |
| VERTEX PHARMACEUTICALS | 376,20 | +0,09 % | If You Invested $100 In Vertex Pharmaceuticals Stock 20 Years Ago, You Would Have This Much Today |
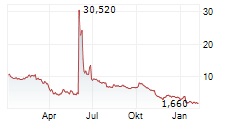
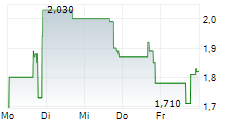
LYRA THERAPEUTICS INC gehört der Branche Pharma an. Der aktuelle Kurs liegt bei 0,495 Euro und damit 0,00 % im Plus.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu LYRA THERAPEUTICS INC finden Sie auf Wallstreet Online.