- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| Fr | FDA grants priority review for Sanofi's venglustat NDA | 24 | Pharmaceutical Technology | ||
| Do | November date set for FDA decision on Sanofi's Gaucher drug | 7 | pharmaphorum | ||
| Do | Sanofi Gets FDA Priority Review For Venglustat In Type 3 Gaucher Disease | 347 | AFX News | PARIS (dpa-AFX) - Sanofi SA (SNY) on Thursday said the US Food and Drug Administration (FDA) has granted priority review to the new drug application for venglustat for the treatment of type... ► Artikel lesen | |
| SANOFI SA CDR Aktie jetzt für 0€ handeln | |||||
| Do | Sanofi: Sanofi's venglustat accepted for priority review in the US to treat type 3 Gaucher disease | 558 | GlobeNewswire (Europe) | Sanofi's venglustat accepted for priority review in the US to treat type 3 Gaucher disease
Paris, May 28, 2026. The US Food and Drug Administration (FDA) has granted priority review to the new drug... ► Artikel lesen | |
| Mi | Sanofi is building its own AI ecosystem to give the French pharma giant an edge | 3 | Fortune | ||
| Mi | Sanofi - 6-K, Report of foreign issuer | 4 | SEC Filings | ||
| 19.05. | Sanofi: Information concerning the total number of voting rights and shares - April 2026 | 42 | GlobeNewswire (USA) | ||
| 19.05. | Sanofi says drug for rare disease AATD tops standard care | 17 | pharmaphorum | ||
| 19.05. | Sanofi Reports Positive Phase 2 Data For Efdoralprin Alfa In AATD-Related Emphysema | 570 | AFX News | PARIS (dpa-AFX) - Sanofi SA (SNY) reported positive data from the Phase 2 ElevAATe study of efdoralprin alfa in adult patients with alpha-1 antitrypsin deficiency-related emphysema.Alpha-1 Antitrypsin... ► Artikel lesen | |
| 18.05. | Sanofi and Wave post AATD updates in race to end genetic disease's 40-year new treatment drought | 5 | FierceBiotech | ||
| 18.05. | Sanofi-Medikament zeigt in Studie zu seltener Krankheit Überlegenheit gegenüber Standardtherapie | 47 | Investing.com Deutsch | ||
| 18.05. | Sanofi drug shows superiority over standard therapy in rare disease | 11 | Investing.com | ||
| 18.05. | Sanofi: ATS: phase 2 data demonstrate the superiority of efdoralprin alfa over a standard-of-care augmentation therapy in achieving higher fAAT levels in AATD | 491 | GlobeNewswire (Europe) | ATS: phase 2 data demonstrate the superiority of efdoralprin alfa over a standard-of-care augmentation therapy in achieving higher fAAT levels in AATD
Data from the head-to-head study show efdoralprin... ► Artikel lesen | |
| 18.05. | Entwicklung von neuen Medikamenten: Wie die Durchführung klinischer Studien bei Sanofi koordiniert wird | 623 | news aktuell | Frankfurt/Main (ots) - Klinische Studien sind mit der wichtigste Schritt bei der Entwicklung von Medikamenten. Mit ihnen werden neue Wirkstoffe unter realen Bedingungen getestet. Die genau geplanten... ► Artikel lesen | |
| 12.05. | Bicara Therapeutics hires Replimune, Sanofi alum as chief commercial officer | 36 | FiercePharma | ||
| 12.05. | Sanofi-Aktie: Jetzt günstig genug? | 1.230 | sharedeals.de | Die Aktie von Sanofi geriet in den letzten Monaten deutlich unter Druck. Gleichzeitig mehren sich jedoch die Anzeichen, dass der Pharmakonzern operativ vor einer deutlich stärkeren Wachstumsphase steht.... ► Artikel lesen | |
| 06.05. | Sanofi asks to pull Tzield bid from FDA's controversial CNPV program: report | 58 | FiercePharma | ||
| 06.05. | Dividendenbekanntmachungen (06.05.2026) | 9.561 | wallstreetONLINE (Dividenden) | Unternehmen ISIN-Code Dividende (Währung) Dividende (EUR) ACCELLERON INDUSTRIES AG CH1169360919 1,5 CHF 1,6376 EUR ALSEA SAB DE CV MXP001391012 1 MXN 0,0492 EUR ALZCHEM GROUP AG DE000A2YNT30 - 2... ► Artikel lesen | |
| 05.05. | Sanofi expands AI capabilities, investing $294M to scale Toronto hub | 28 | FiercePharma | ||
| 05.05. | Sanofi - 6-K, Report of foreign issuer | 7 | SEC Filings |
1 2 3 4 5 Weiter >>
566 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BAYER | 33,470 | -4,73 % | Bayer-Aktie: Das könnte brandgefährlich werden | Die Bayer-Aktie pendelte in den letzten drei Monaten in einem Seitwärtstrend zwischen 36 und 41 € auf und ab. Doch nun könnte eine Nachricht die Aktie aus diesem Trend nach unten reißen. Wie sollten... ► Artikel lesen | |
| MERCK KGAA | 130,80 | 0,00 % | DAX-Check LIVE: Delivery Hero, Infineon, Merck, Nordex, Secunet, Siemens im Fokus | Der deutsche Aktienmarkt hat den Pfingstmontag mit einem fulminanten Kurssprung begangen. Der DAX startete mit Gewinnen, übersprang direkt die wichtige Marke von 25.000 Punkten und kletterte im Verlauf... ► Artikel lesen | |
| NOVO NORDISK | 37,750 | -1,05 % | Vorsicht bei BioNTech! Novo Nordisk gibt Gas! Kaufchance bei Vidac Pharma Aktie! | Kaufstimmung bei Novo Nordisk. Die Aktie des dänischen Pharmagiganten scheint den seit Sommer 2024 begonnenen Abwärtstrend zu verlassen. Anleger reagieren auf positive Meldungen. Bei Vidac Pharma ist... ► Artikel lesen | |
| PFIZER | 22,020 | -0,07 % | Pfizer: Gedämpfte Erwartungen in der Krebsforschung lasten auf der Aktie | Pfizer bleibt unter Druck, denn Barclays sieht trotz laufender Krebsstudien kaum Chancen auf eine Kursrally. Die Analysten halten das Potenzial der Onkologie-Pipeline für begrenzt und bestätigen ihr... ► Artikel lesen | |
| ELI LILLY | 929,40 | -0,10 % | HBM Healthcare Investments AG: Eli Lilly übernimmt HBM-Portfoliounternehmen Curevo für bis zu USD 1.5 Milliarden | HBM Healthcare Investments AG
/ Schlagwort(e): Ankauf
Eli Lilly übernimmt HBM-Portfoliounternehmen Curevo für bis zu USD 1.5 Milliarden
26.05.2026 / 13:10 CET/CEST
Die... ► Artikel lesen | |
| BRIGHT MINDS BIOSCIENCES | 81,47 | +0,56 % | Bright Minds Biosciences Announces Closing of US$175 Million Public Offering | NEW YORK, Jan. 09, 2026 (GLOBE NEWSWIRE) -- Bright Minds Biosciences Inc. (CSE: DRUG) (NASDAQ: DRUG) ("Bright Minds" or the "Company") today closed its recently announced public offering of 1,945... ► Artikel lesen | |
| ETON PHARMACEUTICALS | 28,680 | 0,00 % | Eton Pharmaceuticals Expands Rare Disease Portfolio Through Agreement for U.S. Commercialization Rights to IMPAVIDO (miltefosine) | IMPAVIDO is the first and only FDA-approved oral therapy for visceral, cutaneous, and mucosal leishmaniasis caused by specific Leishmania speciesLeishmaniasis is a rare but potentially life-threatening... ► Artikel lesen | |
| VERTEX PHARMACEUTICALS | 375,95 | -0,27 % | Vertex: FDA Accepts BLA Submission For Povetacicept; Assigns PDUFA Target Action Date Of Nov. 30 | CAMBRIDGE (MASSACHUSETTS) (dpa-AFX) - Vertex Pharmaceuticals Incorporated (VRTX) announced the FDA has accepted its Biologics License Application submission for povetacicept, an investigational... ► Artikel lesen | |
| GENERATE BIOMEDICINES | 12,970 | 0,00 % | Generate:Biomedicines: Generate Biomedicines, Inc. Reports First Quarter 2026 Financial Results and Provides Business Update | SOLAIRIA-1 AND SOLAIRIA-2 Phase 3 clinical trials for GB-0895 (anti-TSLP) continue to advance
Clinical trial sites activated for GB-4362 (MMAE neutralizer); first... ► Artikel lesen | |
| NOVARTIS | 125,94 | +0,27 % | Mesoblast: Während Amgen und Novo Nordisk den Biotech-Hype treiben, arbeitet dieser Zelltherapie-Player am operativen Durchbruch | ||
| ABBOTT LABORATORIES | 75,46 | +0,05 % | EQS-News: Abbott erhält CE-Kennzeichnung für die weltweit erste duale Glukose-Keton-Sensortechnologie für Menschen mit Diabetes | EQS-News: Abbott
/ Schlagwort(e): Expansion/Sonstiges
Abbott erhält CE-Kennzeichnung für die weltweit erste duale Glukose-Keton-Sensortechnologie für Menschen mit Diabetes
27.05.2026... ► Artikel lesen | |
| ABBVIE | 183,30 | +0,11 % | AbbVie Announces European Commission Approval of AQUIPTA (atogepant) for the Acute Treatment of Migraine in Adults | AQUIPTA (atogepant) is now approved in the European Union for the acute treatment of migraine in adults, with or without aura, and for the prophylaxis of migraine in adults who have at least... ► Artikel lesen | |
| MERCK & CO | 98,68 | -0,35 % | Ihre wichtigsten Termine: Alle Analysten-Augen auf: Aroundtown, Kingfisher sowie Merck & Co | © Foto: © 2023 Merck & CoGut geplant in den Tag. Mit dem wO-Tagesausblick haben Sie die wichtigsten Termine im Blick und starten bestens vorbereitet in den neuen Handelstag.Unternehmenstermine 06:45... ► Artikel lesen | |
| GSK | 21,170 | -0,38 % | GSK erzielt teilweisen Therapieerfolg mit Hepatitis-B-Wirkstoff | DJ GSK erzielt teilweisen Therapieerfolg mit Hepatitis-B-Wirkstoff
Von Adria Calatayud
DOW JONES--GSK hat mit einem Medikamentenkandidaten gegen Hepatitis B einen teilweisen Therapieerfolg... ► Artikel lesen | |
| ZOETIS | 66,74 | +0,09 % | Argus downgrades Zoetis stock rating on competition, margin pressure |
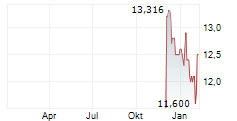
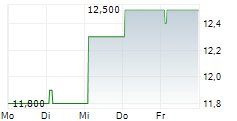
SANOFI SA CDR gehört der Branche Pharma an. Die relative Kursveränderung beträgt aktuell 0,00 % bei einem Aktienkurs von 11,300 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu SANOFI SA CDR finden Sie auf Wallstreet Online.