| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
| 1,796 | 1,835 | 04.06. | |
| 1,788 | 1,843 | 04.06. |
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 29.05. | FOSUN PHARMA Innovative Oncology Immunotherapy FXB0871 Completes First Participant Dosing in China Phase I Trial | 7 | AASTOCKS | ||
| 29.05. | FOSUN PHARMA, GENRIX Sign Strategic Cooperation Framework Agreement | 9 | AASTOCKS | ||
| 21.05. | FOSUN PHARMA (02196): OVERSEAS REGULATORY ANNOUNCEMENT - ANNOUNCEMENT IN RELATION TO THE APPROVAL FOR DRUG REGISTRATION OF A SUBSIDIARY | 29 | HKEx | ||
| 14.05. | Fosun Pharma, AriBio Sign Exclusive Global Option Agreement | 18 | Contract Pharma | ||
| 14.05. | Fosun Pharma to Pay Up to USD240 Million More for Global Rights to AriBio's Early Alzheimer's Drug | 3 | Yicai Global | ||
| 14.05. | FOSUN PHARMA Granted Exclusive Commercialization Option for Alzheimer's Disease Therapy | 12 | AASTOCKS | ||
| 13.05. | FOSUN PHARMA (02196): OVERSEAS REGULATORY ANNOUNCEMENT - ANNOUNCEMENT IN RELATION TO THE ACCEPTANCE OF A SUBSIDIARY'S DRUG REGISTRATION APPLICATION | 3 | HKEx | ||
| SHANGHAI FOSUN PHARMACEUTICAL Aktie jetzt für 0€ handeln | |||||
| 11.05. | FOSUN PHARMA (02196): NOTIFICATION LETTER AND REQUEST FORM TO NON-REGISTERED SHAREHOLDERS | 2 | HKEx | ||
| 11.05. | FOSUN PHARMA (02196): NOTIFICATION LETTER AND REPLY FORM TO REGISTERED SHAREHOLDERS | 10 | HKEx | ||
| 11.05. | FOSUN PHARMA (02196): FORM OF PROXY FOR HOLDERS OF H SHARES FOR THE 2026 SECOND CLASS MEETING OF HOLDERS OF H SHARES TO BE HELD ON TUESDAY, 16 JUNE 2026 | 1 | HKEx | ||
| 11.05. | FOSUN PHARMA (02196): FORM OF PROXY FOR HOLDERS OF H SHARES FOR THE ANNUAL GENERAL MEETING TO BE HELD ON TUESDAY, 16 JUNE 2026 | 1 | HKEx | ||
| 11.05. | FOSUN PHARMA (02196): NOTICE OF 2026 SECOND H SHAREHOLDERS CLASS MEETING | 2 | HKEx | ||
| 11.05. | FOSUN PHARMA (02196): NOTICE OF ANNUAL GENERAL MEETING | 2 | HKEx | ||
| 11.05. | FOSUN PHARMA (02196): ANNUAL GENERAL MEETING; REMUNERATION MANAGEMENT SYSTEM FOR DIRECTORS AND SENIOR MANAGEMENT; ENTRUSTED LOANS/BORROWINGS QUOTA; AUTHORISATION ... | 3 | HKEx | ||
| 08.05. | FOSUN PHARMA (02196): OVERSEAS REGULATORY ANNOUNCEMENT - ANNOUNCEMENT IN RELATION TO THE APPROVAL FOR DRUG REGISTRATION OF A SUBSIDIARY | 2 | HKEx | ||
| 30.04. | FOSUN PHARMA (02196): OVERSEAS REGULATORY ANNOUNCEMENT - ANNOUNCEMENT IN RELATION TO THE APPROVAL FOR DRUG REGISTRATION OF A SUBSIDIARY | 3 | HKEx | ||
| 28.04. | Shanghai Fosun Pharmaceutical (Group) Co Ltd Q1 Income Rises | 4 | RTTNews | ||
| 28.04. | Fosun Pharma Announces Q1 2026 Results: Net Profit Attributable to Shareholders After Deducting Non-Recurring Gains and Losses Increased by 21.96% YoY, With Strong Pipeline Execution | 665 | PR Newswire | SHANGHAI, April 28, 2026 /PRNewswire/ -- On 28 April, Fosun Pharma ("the Company", stock code: 600196.SH; 02196.HK) announced its results for the first quarter... ► Artikel lesen | |
| 28.04. | FOSUN PHARMA (02196): 2026 FIRST QUARTERLY REPORT | 1 | HKEx | ||
| 28.04. | Arcera Life Sciences and Fosun Pharma launch long-term collaboration framework | 218 | PR Newswire | Strategic framework established for regional and global licensing, frontier technology incubation, and strategic collaboration in neurodegenerative diseases
Joint R&D aligned with... ► Artikel lesen |
1 2 3 4 Weiter >>
78 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BAYER | 35,560 | +2,48 % | Bayer, MustGrow Biologics, K+S - die geheime Gewinnformel hinter der biologischen Dünger-Revolution | Erstellt und veröffentlicht im Auftrag von MustGrow Biologics Corp. Die globale Düngemittelbranche durchläuft ihren bedeutendsten Strukturwandel seit Generationen. Steigende Weltbevölkerung, technologische... ► Artikel lesen | |
| NOVO NORDISK | 37,535 | -0,31 % | Novo Nordisk Aktie: Jetzt oder nie! | © Foto: SHVETS production auf Pexels (Symbolbild)Wer Novo Nordisk in den letzten Monaten beobachtet hat, kennt das Auf und Ab dieser Aktie. Der dänische Pharmakonzern, jahrelang gefeierter Star der... ► Artikel lesen | |
| PFIZER | 22,150 | +0,07 % | Pfizer: Gedämpfte Erwartungen in der Krebsforschung lasten auf der Aktie | Pfizer bleibt unter Druck, denn Barclays sieht trotz laufender Krebsstudien kaum Chancen auf eine Kursrally. Die Analysten halten das Potenzial der Onkologie-Pipeline für begrenzt und bestätigen ihr... ► Artikel lesen | |
| NOVARTIS | 127,02 | -0,27 % | Aktien Europa: Leichte Verluste - fragile Waffenruhe im Iran mahnt zur Vorsicht | PARIS/LONDON/ZÜRICH (dpa-AFX) - Die europäischen Aktienmärkte haben am Donnerstag leicht nachgegeben. Die fragile Waffenruhe im Iran-Krieg sorgte für Zurückhaltung. So hatte das US-Militär nahe der... ► Artikel lesen | |
| MERCK KGAA | 139,50 | +4,49 % | Mesoblast: Während Amgen und Novo Nordisk den Biotech-Hype treiben, arbeitet dieser Zelltherapie-Player am operativen Durchbruch | ||
| GILEAD SCIENCES | 111,40 | +0,11 % | Während Merck, Amgen, Novo Nordisk und Fresenius Milliarden bewegen, wächst die Fantasie im Zelltherapie-Sektor | ||
| AURORA CANNABIS | 3,036 | 0,00 % | Aurora Deepens its Impact for Veterans Across Canada | ||
| SANOFI | 76,62 | -0,08 % | DEUTSCHE BANK RESEARCH stuft SANOFI auf 'Buy' | FRANKFURT (dpa-AFX Analyser) - Deutsche Bank Research hat die Einstufung für Sanofi mit einem Kursziel von 100 Euro auf "Buy" belassen. In den Gesprächen mit den Franzosen sei es vor allem um den Umbruch... ► Artikel lesen | |
| GSK | 21,940 | +2,48 % | GSK erzielt teilweisen Therapieerfolg mit Hepatitis-B-Wirkstoff | DJ GSK erzielt teilweisen Therapieerfolg mit Hepatitis-B-Wirkstoff
Von Adria Calatayud
DOW JONES--GSK hat mit einem Medikamentenkandidaten gegen Hepatitis B einen teilweisen Therapieerfolg... ► Artikel lesen | |
| ABBVIE | 193,75 | +0,03 % | Drei Qualitätswerte im Fokus: Was der "Quant Growth And Income"-Ansatz jetzt über Microsoft, Visa und AbbVie verrät | Microsoft, Visa und AbbVie stehen im Zentrum eines quantitativen Wachstums- und Einkommensansatzes, den Seeking Alpha aktuell transparent macht. Der Einblick in das "Quant Growth And Income"-Portfolio... ► Artikel lesen | |
| ROCHE | 356,03 | +0,08 % | Verlagerung in die USA: Roche, Novartis, Ypsomed: Schweizer Firmenparadies North Carolina | ||
| CANOPY GROWTH | 0,906 | +0,87 % | Canopy Growth Corporation: Canopy Growth Relaunches Tweed Brand in Germany with New MTL Cannabis Strain Lineup, Marking First International Release following Acquisition | Pablo's Revenge, Dante'z Inferno, and Frost'd Flakes launched under Tweed brand Up to five additional strains expected to follow in June 2026 Company targeting rapid growth in Germany's medical... ► Artikel lesen | |
| STADA ARZNEIMITTEL | - | - | Enzymatica AB: Quarterly report Q1/2026 - Enzymatica strengthens international position through STADA agreement | First quarter
Net sales amounted to SEK 11.4 million (12.3).
Operating result amounted to SEK -13.6.million (-18.0)
Earnings per share, before... ► Artikel lesen | |
| ELI LILLY | 968,50 | -0,06 % | LimmaTech Biologics wird von Lilly für bis zu 780 Millionen US-Dollar übernommen | Schlieren - LimmaTech Biologics AG, ein Schweizer Biotech-Unternehmen im klinischen Stadium, das Impfstoffe zur Prävention lebensbedrohlicher Krankheiten entwickelt, gab den Abschluss einer endgültigen... ► Artikel lesen | |
| MERCK & CO | 103,26 | -0,31 % | Moderna, Inc.: Moderna and Merck Present 5-Year Data for Intismeran Autogene in Combination With KEYTRUDA in Patients With High-Risk Stage III/IV Melanoma Following Complete Resection at the 2026 ASCO Annual Meeting | At a median 5-year (60.3 months) planned follow-up of the Phase 2b KEYNOTE-942/mRNA-4157-P201 study, intismeran autogene in combination with KEYTRUDA demonstrated a 49% reduction in the risk of recurrence... ► Artikel lesen |
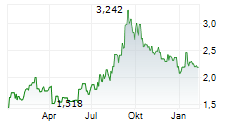
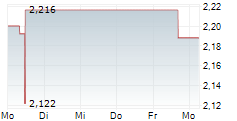
Das Unternehmen SHANGHAI FOSUN PHARMACEUTICAL GROUP CO LTD kann der Branche Pharma zugeordnet werden. Die relative Kursveränderung beträgt aktuell -1,93 % bei einem Aktienkurs von 1,780 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu SHANGHAI FOSUN PHARMACEUTICAL GROUP CO LTD finden Sie auf Wallstreet Online.