- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 13.05. | Aprea Therapeutics GAAP EPS of -$0.22 beats by $0.01 | 2 | Seeking Alpha | ||
| 13.05. | Aprea Therapeutics, Inc. - 10-Q, Quarterly Report | 2 | SEC Filings | ||
| 13.05. | Aprea Therapeutics Reports First Quarter 2026 Financial Results and Provides a Corporate Update | 2.285 | GlobeNewswire (Europe) | Oversubscribed $30 million private placement closed, with proceeds expected to support ongoing development of APR-1051Two partial responses observed with continued encouraging tolerability in the... ► Artikel lesen | |
| 13.05. | Aprea Therapeutics, Inc. - 8-K, Current Report | 4 | SEC Filings | ||
| 07.05. | This Aprea Therapeutics Analyst Begins Coverage On A Bullish Note; Here Are Top 3 Initiations For Thursday | 2 | Benzinga.com | ||
| 07.05. | Oppenheimer initiates Aprea stock with Outperform on WEE1 inhibitor potential | 10 | Investing.com | ||
| 21.04. | Aprea Therapeutics abstract accepted at ASCO meeting in May | 1 | Investing.com | ||
| APREA THERAPEUTICS Aktie jetzt für 0€ handeln | |||||
| 21.04. | Aprea Therapeutics to Provide Clinical Update on ACESOT-1051 Phase 1 Trial Evaluating WEE1 Inhibitor, APR-1051, at ASCO 2026 Annual Meeting | 2 | GlobeNewswire (USA) | ||
| 01.04. | Aprea Therapeutics closes $30 million private placement | 3 | Investing.com | ||
| 01.04. | Aprea Therapeutics, Inc. - 8-K, Current Report | - | SEC Filings | ||
| 30.03. | Aprea reports confirmed tumor response in WEE1 inhibitor trial | 2 | Investing.com | ||
| 30.03. | Aprea Therapeutics sichert sich 30 Millionen US-Dollar durch Privatplatzierung | 1 | Investing.com Deutsch | ||
| 30.03. | Aprea Therapeutics Announces Oversubscribed $30 Million Private Placement | 1 | GlobeNewswire (USA) | ||
| 16.03. | Aprea Therapeutics, Inc. - S-8, Securities to be offered to employees in employee benefit plans | 1 | SEC Filings | ||
| 16.03. | H.C. Wainwright reiterates Aprea stock rating, keeps $4 target | 2 | Investing.com | ||
| 16.03. | Aprea Therapeutics GAAP EPS of -$0.32 beats by $0.07 | 3 | Seeking Alpha | ||
| 16.03. | Aprea Therapeutics, Inc. - 10-K, Annual Report | 4 | SEC Filings | ||
| 16.03. | Aprea Therapeutics, Inc. - 8-K, Current Report | 1 | SEC Filings | ||
| 16.03. | Aprea Therapeutics Reports Fourth Quarter and Full Year 2025 Financial Results and Provides a Corporate Update | 1.186 | GlobeNewswire (Europe) | Early clinical proof-of-concept for WEE1 inhibitor APR-1051 in the ongoing ACESOT-1051 trial with two partial responders at first scan in endometrial cancer patients with PPP2R1A mutationClinical... ► Artikel lesen | |
| 18.02. | Aprea Therapeutics Advanced Cancer Trial Shows Promising Results, Stock Soars | 4 | Benzinga.com |
1 2 3 Weiter >>
46 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| EVOTEC | 4,932 | -3,58 % | EQS-News: Evotec SE: Evotec ernennt Rui Wang zum Head of Global In Silico & AI | EQS-News: Evotec SE
/ Schlagwort(e): Sonstiges
Evotec ernennt Rui Wang zum Head of Global In Silico & AI
02.06.2026 / 07:30 CET/CEST
Für den Inhalt der Mitteilung ist... ► Artikel lesen | |
| BB BIOTECH | 45,850 | -0,86 % | BB Biotech: "Wachstumsstarke Mid- und Small Caps bilden das Rückgrat unseres Portfolios": Mehr Einzelwerte und ... | ||
| VALNEVA | 2,331 | -0,64 % | Valneva im Höhenflug: Das Signal das die meisten Anleger noch nicht kennen - Meine Einschätzung für die neue Börsenwoche | ||
| BIOGEN | 171,56 | +1,19 % | Biogen's Salanersen Wins FDA Breakthrough Therapy Designation For Spinal Muscular Atrophy Treatment | WESTON (dpa-AFX) - Thursday, Biogen Inc. (BIIB) announced that the U.S. Food and Drug Administration has granted salanersen Breakthrough Therapy Designation for the treatment of spinal muscular... ► Artikel lesen | |
| ILLUMINA | 139,80 | -0,70 % | ILMN Stock Jumps 98.7% in a Year: What's Powering the Surge? | ||
| ABIVAX | 87,00 | -0,63 % | Abivax mit Daten: Erst Rakete, dann Absturz - erhöht Obefazimod das Krebsrisiko? | Die Aktie von Abivax hat am Montagabend genau die Nachricht geliefert, auf die Investoren seit Monaten gewartet hatten. Das französische Biotech-Unternehmen präsentierte herausragende Topline-Ergebnisse... ► Artikel lesen | |
| GINKGO BIOWORKS | 6,904 | -2,90 % | Ginkgo Bioworks Reports First Quarter 2026 Financial Results, Completes Divestiture of Biosecurity and Continues to Scale Autonomous Lab | Ginkgo provides an update on its first quarter financial results following the divestiture of its Biosecurity business
BOSTON, May 7, 2026 /PRNewswire/ -- Ginkgo... ► Artikel lesen | |
| ONCO-INNOVATIONS | 0,520 | -2,80 % | Onco-Innovations Limited: Onco-Innovations Signs LOI with Nanosoft Polymers to Advance Polymer and Drug Delivery Development for the ONC010 Program | VANCOUVER, BC / ACCESS Newswire / June 5, 2026 / Onco-Innovations Limited (CBOE CA:ONCO)(OTCQB:ONNVF)(Frankfurt:W1H, WKN: A3EKSZ) ("Onco" or the "Company") is pleased to announce that it has entered... ► Artikel lesen | |
| PRAXIS PRECISION MEDICINES | 254,00 | 0,00 % | Praxis Precision Medicines, Inc.: Praxis Precision Medicines Provides Vormatrigine Program Update | POWER1 Study in highly refractory patients with focal onset seizures did not meet its primary success measure Secondary measure, the 50% response rate, was met and seizure reduction during the second... ► Artikel lesen | |
| EDGEWISE THERAPEUTICS | 35,590 | 0,00 % | Servier to Acquire Edgewise Therapeutics' Muscular Dystrophy Business | Acquisition supports Servier's strategic ambition in rare neurology for patients with high unmet medical needs.Transaction includes sevasemten, a late-stage investigational treatment for... ► Artikel lesen | |
| JADE BIOSCIENCES | 16,940 | 0,00 % | Jade Biosciences Announces Positive Interim Phase 1 Results for JADE101 Demonstrating Potential Best-in-Class Profile and Every-12-Week Dosing in IgA Nephropathy | ~70% IgA reductions sustained at 12 weeks after a single dose of JADE101>70% IgA reductions projected at steady state with a single subcutaneous maintenance injection every 12 weeksIgA-lowering effect... ► Artikel lesen | |
| QIAGEN | 31,920 | -2,18 % | DEUTSCHE BANK RESEARCH stuft QIAGEN NV auf 'Buy' | FRANKFURT (dpa-AFX Analyser) - Deutsche Bank Research hat Qiagen mit einem Kursziel von 43 US-Dollar auf "Buy" belassen. Das Feedback aus Gesprächen mit etlichen US-Laborexperten deute darauf hin, dass... ► Artikel lesen | |
| APOGEE THERAPEUTICS | 86,86 | -0,07 % | Weekly Buzz: AstraZeneca, JNJ Get FDA Nod; Eli Lilly To Acquire Curevo; APGE Advances Zumilokibart | NEW BRUNSWICK (dpa-AFX) - This week in the biotech space, there were regulatory approvals across the U.S. and Europe, collaborations, acquisitions, and positive data readouts spanning multiple... ► Artikel lesen | |
| ADMA BIOLOGICS | 7,970 | 0,00 % | ADMA BIOLOGICS, INC. - 8-K, Current Report | ||
| SUMMIT THERAPEUTICS | 14,755 | -0,10 % | All You Need to Know About Summit Therapeutics (SMMT) Rating Upgrade to Buy |
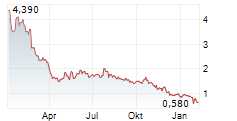
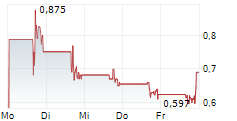
Das Unternehmen APREA THERAPEUTICS INC kann der Branche Biotechnologie zugeordnet werden. Die relative Kursveränderung beträgt aktuell 0,00 % bei einem Aktienkurs von 0,741 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu APREA THERAPEUTICS INC finden Sie auf Wallstreet Online.