- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 10.04. | The observation status for Guard Therapeutics International AB (publ) is updated | 239 | GlobeNewswire | On December 11, 2025, the shares in Guard Therapeutics International AB (publ) (the "Company") were given observation status with reference to information that the Company's board of directors had... ► Artikel lesen | |
| 09.04. | Guard Therapeutics International AB: Guard Therapeutics provides information regarding observation status on Nasdaq First North Growth Market | 168 | GlobeNewswire (Europe) | Guard Therapeutics International AB (publ) ("Guard Therapeutics" or the "Company") today announces that Nasdaq has informed the Company that it currently does not fulfil the ongoing listing requirement... ► Artikel lesen | |
| 31.03. | Guard Therapeutics International AB: Guard Therapeutics provides update on strategic review | 206 | GlobeNewswire (Europe) | Guard Therapeutics International AB (publ) ("Guard Therapeutics" or the "Company") hereby provides an update regarding the strategic review that was announced in December 2025.
The Company has now completed... ► Artikel lesen | |
| 21.02. | Guard Therapeutics International AB Q4 Loss Decreases | 7 | RTTNews | ||
| 05.02. | Guard Therapeutics International AB: Guard Therapeutics Announces Changes to the Company's Management Team | 348 | GlobeNewswire (Europe) | Guard Therapeutics (publ) today announces changes to the company's management team, in light of the conclusion of the clinical program for the drug candidate RMC-035 and the ongoing strategic review... ► Artikel lesen | |
| GUARD THERAPEUTICS INTERNATIONAL Aktie jetzt für 0€ handeln | |||||
| 05.02. | Guard Therapeutics International AB: Guard Therapeutics Provides Status Update Regarding Ongoing Strategic Review | 272 | GlobeNewswire (Europe) | Guard Therapeutics International AB (publ) ("Guard Therapeutics" or the "Company") hereby provides a status update regarding the strategic review that the Company announced through a press release on... ► Artikel lesen | |
| 15.01. | Guard Therapeutics International AB: Guard Therapeutics Partners in EU Consortium For Alport Syndrome | 490 | GlobeNewswire (Europe) | Guard Therapeutics (publ) today announced its participation as a partner in a newly funded European research consortium under the 2025 Joint Transnational Call from the European Rare Diseases Research... ► Artikel lesen | |
| 11.12.25 | Guard Therapeutics International AB (publ) receives observation status | 274 | GlobeNewswire | On December 4, 2025, Guard Therapeutics International AB (publ) (the "Company") issued a press release with information that the Board of Directors of the Company had resolved to commence an exploratory... ► Artikel lesen | |
| 04.12.25 | Guard Therapeutics International AB: Guard Therapeutics Explores Strategic Alternatives | 258 | GlobeNewswire (Europe) | Guard Therapeutics (publ) ("Guard" or the "Company") today announces that the Board of Directors has resolved to commence an exploratory process regarding strategic alternatives, including identifying... ► Artikel lesen | |
| 04.12.25 | Guard Therapeutics International AB: Guard Therapeutics Provides Assessment of Full POINTER Study Results | 252 | GlobeNewswire (Europe) | Guard Therapeutics (public) ("Guard" or "the Company") today provides an assessment of the full results from the Phase 2b POINTER study evaluating RMC-035 in patients undergoing open-heart surgery.... ► Artikel lesen | |
| 26.10.25 | Guard Therapeutics International AB: Guard Therapeutics Announces Top-Line Results from Phase 2b POINTER Study | 610 | GlobeNewswire (Europe) | Guard Therapeutics (publ) today announced top-line results from the company's Phase 2b POINTER study evaluating the investigational drug candidate RMC-035 as a kidney-protective therapy in patients... ► Artikel lesen | |
| 05.10.25 | Guard Therapeutics International AB: Guard Therapeutics Announces New Positive Efficacy Results from Secondary Analyses of the Phase 2a AKITA Study | 287 | GlobeNewswire (Europe) | Guard Therapeutics (publ) today announced favorable efficacy results from additional analyses of the Phase 2a AKITA study with the investigational drug candidate RMC-035. The previously reported primary... ► Artikel lesen | |
| 11.09.25 | Guard Therapeutics International AB: Guard Therapeutics announces completion of data collection in Phase 2b POINTER study | 807 | GlobeNewswire (Europe) | Guard Therapeutics (publ) today announced that the last patient in its ongoing Phase 2b clinical trial, POINTER, has successfully completed the final scheduled follow-up visit 90 days after surgery... ► Artikel lesen | |
| 21.08.25 | Guard Therapeutics International AB: Guard Therapeutics publishes interim report January-June 2025 | 274 | GlobeNewswire (Europe) | The quarter marked a key milestone with the completion of patient recruitment for our Phase 2b POINTER study - ahead of schedule. The positive final safety review reinforces confidence in the trial... ► Artikel lesen |
14 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| NOVONESIS | 48,800 | -0,02 % | Novonesis Q1 Net Profit Rises, Net Sales Up 7% Organically; Backs FY26 Outlook | BRUSSELS (dpa-AFX) - Novonesis A/S (NVZMF.PK, NSIS_B.CO, NSIS-B.CO, NVZMF, NSIS-B) Tuesday said that its first quarter net profit rose from last year, helped by good performances by the Food... ► Artikel lesen | |
| GENMAB | 213,60 | -0,42 % | Genmab A/S: Genmab to Highlight Advances Across Its Oncology Portfolio at the 2026 American Society of Clinical Oncology (ASCO) Annual Meeting and the European Hematology Association (EHA) 2026 Congress | Twenty-three abstracts will highlight the versatility and strength of Genmab's portfolio and pipeline, including data from the comprehensive epcoritamab development program, including 4 oral... ► Artikel lesen | |
| ARBUTUS BIOPHARMA | 3,670 | 0,00 % | Arbutus Biopharma Corp - S-8, Securities to be offered to employees in employee benefit plans | ||
| MUSTGROW BIOLOGICS | 0,310 | +3,33 % | Börsenrotation ohne KI! Jetzt 100 % mit MustGrow, Bayer und Novo Nordisk, Achtung bei K+S | Erstellt und veröffentlicht im Auftrag von MustGrow Biologics Corp. Perspektivenwechsel bleibt das Gebot der Stunde. Denn gestern tagte die EZB zur aktuellen Lage bei Inflation und Zinsniveau. Mit einer... ► Artikel lesen | |
| VERICEL | 32,000 | +2,56 % | Vericel Corporation: Vericel Reports First Quarter 2026 Financial Results and Raises Full-Year Financial Guidance | Total Revenue Increased 30% to $68.4 Million, with MACI Revenue Growth of 22% and Burn Care Revenue Growth of 91% Gross Margin of 72% and Adjusted EBITDA Growth of 195% Free Cash Flow of $15.1 Million... ► Artikel lesen | |
| MOLECULIN BIOTECH | 2,470 | 0,00 % | Moleculin Biotech, Inc.: Independent Market Research Finds Strong Physician Intent to Prescribe Moleculin's Annamycin in Relapsed/Refractory AML | ||
| SENSEI BIOTHERAPEUTICS | 12,960 | -0,31 % | SelfCare Sensei, LLC: Wellbeing Educator and Martial Artist Jan Raeder Officially Launches Strive for M.O.R.E., the Book That Found 400 Readers Before It Ever Had a Launch | After three decades on the mat and a master's in wellbeing education, Raeder is done watching people settle for bubble baths when the science offers something betterPOTTERSVILLE, N.Y., June... ► Artikel lesen | |
| BIOXCEL THERAPEUTICS | 0,988 | +2,92 % | BioXcel Therapeutics Provides Business Update and Reports First Quarter 2026 Financial Results | FDA accepted sNDA for IGALMI® use in at-home (outpatient) setting; set Nov. 14, 2026, as PDUFA date Advancing BXCL501 as a potential acute treatment for agitation associated with Alzheimer's dementia... ► Artikel lesen | |
| REGENXBIO | 5,364 | +0,98 % | H.C. Wainwright lowers Regenxbio stock price target on trial data | ||
| AKEBIA | 0,806 | -0,42 % | Akebia Therapeutics, Inc.: Akebia Therapeutics Appoints Biopharmaceutical Leader Philip Vickers, Ph.D. to its Board of Directors | CAMBRIDGE, Mass., April 01, 2026 (GLOBE NEWSWIRE) -- Akebia Therapeutics- , Inc. (Nasdaq: AKBA), a biopharmaceutical company with the purpose to better the lives of people impacted by kidney disease... ► Artikel lesen | |
| ZEVRA THERAPEUTICS | 10,300 | -1,90 % | Zevra Therapeutics Announces Orange Book Listing of U.S. Patent Strengthening Exclusivity for MIPLYFFA | MIPLYFFA patent submitted for Orange Book listing, with patent protection to 2041 Separately filed patent term extension remains pending with the U.S. Patent and Trademark Office BOSTON, June 08,... ► Artikel lesen | |
| FENNEC PHARMACEUTICALS | 7,800 | -4,88 % | Sensofusion stellt seine Luft- und Raumfahrtsparte vor: Swift-Überwachungsflugzeuge und Fennec-Satelliten | Das finnische Unternehmen für Verteidigungstechnologie Sensofusion Oy weitet seine Geschäftstätigkeit vom Boden in die Luft und in die Erdumlaufbahn aus. Ab heute wird die Luft- und Raumfahrtsparte... ► Artikel lesen | |
| CORVUS PHARMACEUTICALS | 9,700 | +0,35 % | Corvus Pharmaceuticals, Inc.: Corvus Pharmaceuticals Appoints Andrew C. Chan, M.D., Ph.D., to Board of Directors | SOUTH SAN FRANCISCO, Calif., April 23, 2026 (GLOBE NEWSWIRE) -- Corvus Pharmaceuticals, Inc. (Nasdaq: CRVS), a clinical-stage biopharmaceutical company, announced today that Andrew C. Chan, M.D.,... ► Artikel lesen | |
| CARTESIAN THERAPEUTICS | 5,200 | 0,00 % | Cartesian Therapeutics, Inc.: Cartesian Therapeutics Secures up to $150 Million of Non-Dilutive Financing from K2 HealthVentures and Expects Topline Data from the Phase 3 AURORA Trial in First Quarter 2027 | Secured up to $150 million of non-dilutive financing from K2 HealthVentures; funding of $50 million from initial tranche extends cash runway into 2028 Financing strengthens Cartesian's financial flexibility... ► Artikel lesen | |
| NANOBIOTIX | 30,520 | 0,00 % | Nanobiotix S.A.: Nanobiotix Provides First Quarter 2026 Operational and Financial Update | Completed a follow-on offering in May 2026 for aggregate gross proceeds of approximately €86.1 million, including issuance of pre-funded warrants to accelerate the development of Company's other platformsNew... ► Artikel lesen |
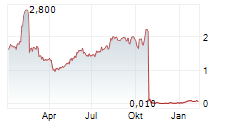
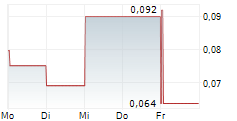
GUARD THERAPEUTICS INTERNATIONAL AB gehört der Branche Biotechnologie an. Der aktuelle Kurs liegt bei 0,028 Euro und damit -12,73 % im Minus.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu GUARD THERAPEUTICS INTERNATIONAL AB finden Sie auf Wallstreet Online.