| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
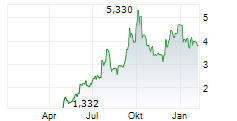
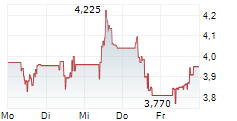
| 4,836 | 4,882 | 29.05. | |
| 4,854 | 4,902 | 29.05. |
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 06.05. | Idorsia Pharmaceuticals Ltd: Shareholders vote in favor of all proposals by the Board at Idorsia's Annual General Meeting 2026 | 734 | GlobeNewswire (Europe) | Allschwil, Switzerland - May 6, 2026At today's Annual General Meeting (AGM) of Idorsia Ltd (SIX: IDIA) held in Basel, Switzerland, shareholders voted convincingly in favor of all proposals by the Board... ► Artikel lesen | |
| 30.04. | Santhera nominiert Ex-Idorsia-Chefin Srishti Gupta als Verwaltungsratsmitglied | 523 | Moneycab | Pratteln - Santhera schlägt der bevorstehenden Generalversammlung am 26. Mai die Wahl von Srishti Gupta in den Verwaltungsrat vor. Diese war von Juli 2025 bis Mitte März 2026 die Chefin von Idorsia.... ► Artikel lesen | |
| IDORSIA Aktie jetzt für 0€ handeln | |||||
| 28.04. | Idorsia sieht sich auf Wachstumskurs nach erstem Quartal | 670 | Moneycab | Allschwil - Das Biotechunternehmen Idorsia ist nach eigenen Aussagen stark in das neue Jahr gestartet. Im ersten Quartal legte der Umsatz mit dem Hoffnungsträger Quviviq (Daridorexant) erneut kräftig... ► Artikel lesen | |
| 28.04. | Idorsia Pharmaceuticals Ltd: Idorsia reports strong Q1 2026 performance with 74% QUVIVIQ sales growth year-on-year | 1.046 | GlobeNewswire (Europe) | Ad hoc announcement pursuant to Art. 53 LR
Preparation is underway to publish the strong pediatric daridorexant data and engage with regulatory authorities on next stepsCo-promotion of QUVIVIQ is... ► Artikel lesen | |
| 10.04. | Idorsia Pharmaceuticals Ltd: Idorsia issues invitation to the 2026 Annual General Meeting of Shareholders | 580 | GlobeNewswire (Europe) | Allschwil, Switzerland - April 10, 2026Idorsia Ltd (SIX: IDIA) today issued the invitation to the upcoming Annual General Meeting (AGM) of Shareholders on behalf of the Board of Directors. The meeting... ► Artikel lesen | |
| 09.04. | Idorsia Pharmaceuticals Ltd: Idorsia welcomes Amer Joseph as new Chief Medical Officer and Head of Global Clinical Development | 676 | GlobeNewswire (Europe) | Idorsia thanks Alberto Gimona - most recently Head of Global Clinical Development and Medical Affairs - and wishes him all the best for a well-deserved retirementAmer Joseph joins Idorsia as EVP, Chief... ► Artikel lesen | |
| 30.03. | Idorsia eyes pediatric insomnia use with midstage trial win for sleep med Quviviq | 9 | FiercePharma | ||
| 30.03. | Idorsia Pharmaceuticals Ltd: Idorsia's daridorexant delivers outstanding results in a Phase 2 study in children with insomnia disorder | 562 | GlobeNewswire (Europe) | Ad hoc announcement pursuant to Art. 53 LR
Phase 2 results show a statistically significant dose-dependent response on total sleep time with clinically meaningful improvements across multiple sleep... ► Artikel lesen | |
| 24.03. | Idorsia Pharmaceuticals Ltd: Idorsia announces the nomination of three candidates for election to the Board of Directors | 844 | GlobeNewswire (Europe) | Idorsia nominates three Board Directors with outstanding pharmaceutical industry, management and financial expertise for election at the Annual General Meeting 2026 on May 6, 2026All Board members -... ► Artikel lesen | |
| 16.03. | Basler Biotechunternehmen: Idorsia-Chefin Gupta tritt nach weniger als einem Jahr zurück | 50 | Schweizer Fernsehen | ||
| 16.03. | Idorsia shares sink after CEO's abrupt departure | 15 | FiercePharma | ||
| 16.03. | Idorsia-Chefin Gupta tritt überraschend zurück - Clozel übernimmt das Zepter | 623 | Moneycab | Allschwil - Paukenschlag bei Idorsia: Nach nicht mal einem Jahr im Amt tritt Srishti Gupta tritt als CEO zurück und scheidet auch aus dem Verwaltungsrat aus. Bis zur Ernennung einer Nachfolge übernimmt... ► Artikel lesen | |
| 16.03. | Idorsia und Pharmalink kooperieren zur Behandlung von Schlafstörungen | 29 | punkt4.info | ||
| 16.03. | Idorsia-Patron Clozel springt operativ wieder ein | 17 | muula.ch | ||
| 16.03. | Idorsia-Chefin Gupta tritt zurück - Clozel wird Interims-CEO | 564 | Moneycab | Allschwil - Paukenschlag bei Idorsia: Nach nicht mal einem Jahr im Amt tritt Srishti Gupta tritt als CEO zurück und scheidet auch aus dem Verwaltungsrat aus. Bis zur Ernennung einer Nachfolge übernimmt... ► Artikel lesen | |
| 16.03. | Idorsia CEO Srishti Gupta To Resign; Search On For Successor | 9 | RTTNews | ||
| 16.03. | Idorsia Pharmaceuticals Ltd: Idorsia announces management change | 630 | GlobeNewswire (Europe) | Ad hoc announcement pursuant to Art. 53 LR
Allschwil, Switzerland - March 16, 2026Idorsia Ltd (SIX: IDIA) announces that Dr Srishti Gupta and the Board of Idorsia have mutually agreed that Srishti... ► Artikel lesen | |
| 11.03. | Idorsia and Pharmalink sign agreement to distribute Quviviq | 25 | Pharmaceutical Technology | ||
| 10.03. | Idorsia Pharmaceuticals Ltd: Idorsia expands QUVIVIQ's global footprint through new partnership with Pharmalink | 1.203 | GlobeNewswire (Europe) | Idorsia and Pharmalink enter into an exclusive distribution and commercialization agreement for QUVIVIQ (daridorexant) in the United Arab Emirates (UAE), Kuwait, Qatar, Oman, and Bahrain
Allschwil... ► Artikel lesen | |
| 26.02. | Idorsia überzeugt in Q4 2025 mit starkem Wachstum in Kernmärkten | 43 | Investing.com Deutsch |
1 2 3 4 Weiter >>
69 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BIONTECH | 82,35 | +0,06 % | Vorsicht bei BioNTech! Novo Nordisk gibt Gas! Kaufchance bei Vidac Pharma Aktie! | Kaufstimmung bei Novo Nordisk. Die Aktie des dänischen Pharmagiganten scheint den seit Sommer 2024 begonnenen Abwärtstrend zu verlassen. Anleger reagieren auf positive Meldungen. Bei Vidac Pharma ist... ► Artikel lesen | |
| EVOTEC | 5,280 | +3,33 % | Shortseller-Positionen aktuell: u.a. Auto1, Evotec, flatexDEGIRO, Heidelberger Druck, LPKF Laser, Scout24, TUI | Wer Aktien leerverkauft, also auf fallende Kurse spekuliert (Shortselling), unterliegt bestimmten Transparenzpflichten. Aktuelle Meldungen über Leerverkäufe geben Aufschluss darüber. Diese Vorgaben... ► Artikel lesen | |
| BB BIOTECH | 47,850 | -1,44 % | Aktien KW 17 Märkte im Würgegriff. Planloser Trump , selbstbewusterer Iran. Lösungen müssen her. News. Rheinmetall. ITM Power. Porsche. The Platform Group. K+S. Bugatti. Circus. Surteco. NanoRepro. BB Biotech. Mensch und Maschine. ATOSS. MHP Hotel | Aktien - letzte Woche warteten die Märkte auf eine Lösung des Iran-Kriegs. Aber die Iraner sehen sich im Vorteil und Trump verlängert "auf unbestimmte Zeit" den Waffenstillstand. Aber die Strasse von... ► Artikel lesen | |
| MEDIGENE | 0,028 | -16,87 % | MEDIGENE AG zeigt nachhaltige Stärke | ||
| QIAGEN | 31,645 | +0,43 % | DEUTSCHE BANK RESEARCH stuft QIAGEN NV auf 'Buy' | FRANKFURT (dpa-AFX Analyser) - Deutsche Bank Research hat die Einstufung für Qiagen mit einem Kursziel von 43 US-Dollar auf "Buy" belassen. Das Interesse der Investoren habe vor allem dem Tuberkulosetest... ► Artikel lesen | |
| MODERNA | 40,455 | +0,10 % | Astellas eyes more cost cuts; FDA panel to debate Moderna vaccine | ||
| PAION | 0,070 | -14,63 % | XFRA MISTRADE ANTRAG IN ISIN DE000A3E5EG5 WURDE STATTGEGEBEN | Dem Mistrade-Antrag wurde stattgegeben. Folgende Geschaefte in der ISIN DE000A3E5EG5, Wertpapier-Name: PAION AG INH O.N., wurden aufgehoben:ISIN Datum Zeit Volumen PreisDE000A3E5EG5 02.02.2026 08:13:40 300 0... ► Artikel lesen | |
| VALNEVA | 2,621 | 0,00 % | Original-Research: Valneva SE (von First Berlin Equity Research GmbH): Buy | Original-Research: Valneva SE - von First Berlin Equity Research GmbH
28.05.2026 / 17:08 CET/CEST
Veröffentlichung einer Research, übermittelt durch EQS News - ein Service der EQS Group.
Für den... ► Artikel lesen | |
| AMGEN | 287,35 | -0,47 % | Mesoblast im Sog der Big Player: Wie Merck, Novartis und Johnson & Johnson den Zelltherapie-Markt vorbereiten | ||
| EPIGENOMICS | 0,880 | 0,00 % | PTA-HV: Epigenomics AG: Einladung zur ordentlichen Hauptversammlung | DJ PTA-HV: Epigenomics AG: Einladung zur ordentlichen Hauptversammlung
Hauptversammlung gemäß -- 121 Abs. 4a AktG
Epigenomics AG: Einladung zur ordentlichen Hauptversammlung
Heidelberg... ► Artikel lesen | |
| NOVAVAX | 9,390 | -0,11 % | Novavax, Inc. (NVAX) Is a Trending Stock: Facts to Know Before Betting on It | ||
| STRYKER | 264,70 | +1,15 % | Stryker bringt das Pangea-Plattensystem auf den Markt und schließt den ersten Eingriff in Europa ab | Stryker (NYSE:SYK), ein weltweit führendes Unternehmen im Bereich Medizintechnik, gab heute die Markteinführung seines Pangea-Plattensystems in Europa bekannt, einer Plattensystem-Plattform zur Behandlung... ► Artikel lesen | |
| BIOGEN | 168,32 | +0,15 % | Biogen investigated by Italian regulator over multiple sclerosis 'market abuse' claims | ||
| BIOFRONTERA | 2,470 | +2,07 % | PTA-News: Biofrontera AG: BIOFRONTERA BERICHTET ÜBER DIE ERGEBNISSE DES ERSTEN QUARTALS 2026 | DJ PTA-News: Biofrontera AG: BIOFRONTERA BERICHTET ÜBER DIE ERGEBNISSE DES ERSTEN QUARTALS 2026
Unternehmensmitteilung für den Kapitalmarkt
Biofrontera AG: BIOFRONTERA BERICHTET ÜBER DIE... ► Artikel lesen | |
| HEIDELBERG PHARMA | 2,710 | +2,26 % | EQS-HV: Heidelberg Pharma AG: Bekanntmachung der Einberufung zur Hauptversammlung am 23.06.2026 in Ladenburg mit dem Ziel der europaweiten Verbreitung gemäß §121 AktG | EQS-News: Heidelberg Pharma AG
/ Bekanntmachung der Einberufung zur Hauptversammlung
Heidelberg Pharma AG: Bekanntmachung der Einberufung zur Hauptversammlung am 23.06.2026 in... ► Artikel lesen |
Das Unternehmen IDORSIA AG kann der Branche Biotechnologie zugeordnet werden. Mit einer aktuellen Kursveränderung von +0,042 (+0,86 %) liegt der Kurs bei 4,920 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu IDORSIA AG finden Sie auf Wallstreet Online.