- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 07.05. | Infant Bacterial Therapeutics: Annual General Meeting of Infant Bacterial Therapeutics | 131 | GlobeNewswire (Europe) | At the Annual General Meeting of Infant Bacterial Therapeutics AB (publ) on May 7, 2026, among other things, the following was resolved: - to adopt the income statement and balance sheet and the consolidated... ► Artikel lesen | |
| 06.05. | Infant Bacterial Therapeutics: Infant Bacterial Therapeutics AB (publ) Interim report January 1 - March 31, 2026 | 192 | GlobeNewswire (Europe) | Message from CEO
Continued constructive dialogue with the FDADuring the period, we continued our interactions with the U.S. Food and Drug Administration (FDA). In February 2026, IBT sent a synopsis... ► Artikel lesen | |
| 06.02. | Infant Bacterial Therapeutics: Infant Bacterial Therapeutics AB (publ) Year-end-report January 1 - December 31, 2025 | 272 | GlobeNewswire (Europe) | Message from CEO
IBT's plan to apply for marketing approval in the US through a so-called accelerated application remains unchanged. The development program for IBP-9414 is "Breakthrough Designated... ► Artikel lesen | |
| 11.12.25 | Infant Bacterial Therapeutics: Infant Bacterial Therapeutics secures partnership with BioConnection for production of Drug Product | 251 | GlobeNewswire (Europe) | Infant Bacterial Therapeutics AB (IBT) whose mission is to develop and commercialize pharmaceuticals for diseases affecting premature babies and BioConnection, a leading pharmaceutical Contract Manufacturing... ► Artikel lesen | |
| 25.11.25 | Infant Bacterial Therapeutics: IBT changes the IBP-9414 pathway for approval following discussions with the FDA | 245 | GlobeNewswire (Europe) | Following today's meeting with the FDA concerning IBT's live biotherapeutic product, IBT has decided to pursue an accelerated approval pathway for IBP-9414.
The FDA's Accelerated Approval Program allows... ► Artikel lesen | |
| 20.11.25 | Infant Bacterial Therapeutics: Infant Bacterial Therapeutics AB secures partnership with Recipharm Advanced Bio for production of drug substance | 621 | GlobeNewswire (Europe) | Infant Bacterial Therapeutics AB (IBT) and Recipharm Advanced Bio, a segment of Recipharm and a leading Contract Development and Manufacturing Organisation (CDMO), announced the initiation of Process... ► Artikel lesen | |
| 13.11.25 | Infant Bacterial Therapeutics: Infant Bacterial Therapeutics AB (publ) Interim report January 1 - September 30, 2025 | 229 | GlobeNewswire (Europe) | Message from CEO
The risk of death decreased by 27% for children who received our drug candidate IBP-9414 compared to placebo in our Phase 3 study. It is difficult for me to put into words the significance... ► Artikel lesen | |
| INFANT BACTERIAL THERAPEUTICS Aktie jetzt für 0€ handeln | |||||
| 13.10.25 | Infant Bacterial Therapeutics: Registration work for IBT's drug candidate IBP-9414 continues in the US and Europe | 257 | GlobeNewswire (Europe) | Communication with the FDA continues, and IBT is focused on obtaining drug approval for IBP-9414 in the US as soon as possible. The next step is a Pre-BLA (Biologics License Application) meeting with... ► Artikel lesen | |
| 19.09.25 | XFRA NEW INSTRUMENTS AVAILABLE ON 19.09.2025 | 1.061 | Xetra Newsboard | The following instruments on XETRA do have their first trading 19.09.2025 Die folgenden Instrumente in XETRA haben ihren ersten Handelstag 19.09.2025
Aktien
1 AU000000EOS8 Electro Optic Systems... ► Artikel lesen | |
| 20.08.25 | Infant Bacterial Therapeutics: Infant Bacterial Therapeutics AB (publ) Interim report January 1 - June 30, 2025 | 261 | GlobeNewswire (Europe) | Message from CEO
IBT continues preparations for the launch of IBP-9414, a drug that can prevent the serious medical consequences of NEC in premature infants. These consequences can be divided into three... ► Artikel lesen |
10 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| SCHOTT PHARMA | 17,620 | +2,20 % | SCHOTT PHARMA AG & CO KGAA: Stabilität als Signal | ||
| XLIFE SCIENCES | 25,000 | +9,65 % | Xlife-Portfoliounternehmen Veraxa stärkt Finanzierung vor IPO | ||
| ALK-ABELLO | 35,140 | +0,51 % | ALK Abello: Report on transactions with ALK-Abelló A/S B shares and associated securities by managerial staff | ||
| POLYPEPTIDE GROUP | 41,200 | +0,49 % | Aktien-Report-Tipp enthüllt: "Schaufelhersteller" PolyPeptide im Übernahme-Visier - neue Chance! | Peptide nehmen eine Schlüsselrolle bei der industriellen Skalierung moderner Stoffwechseltherapien ein. Mit dem Siegeszug der GLP-1-Präparate wächst der Bedarf an den "Schaufeln". Und die an der Schweizer... ► Artikel lesen | |
| BAYER | 33,370 | -5,01 % | Märkte am Morgen: Delivery Hero, MTU, Lufthansa, TUI, Airbus, Bayer, Space X | Nach der außergewöhnlichen Feiertagskonstellation - Pfingstmontag mit Handel in Deutschland, Memorial Day ohne Handel in den USA - tritt der DAX heute leicht auf die Bremse (-0,3 % erwartet). Gestern... ► Artikel lesen | |
| MERCK KGAA | 130,80 | 0,00 % | DAX-Check LIVE: Delivery Hero, Infineon, Merck, Nordex, Secunet, Siemens im Fokus | Der deutsche Aktienmarkt hat den Pfingstmontag mit einem fulminanten Kurssprung begangen. Der DAX startete mit Gewinnen, übersprang direkt die wichtige Marke von 25.000 Punkten und kletterte im Verlauf... ► Artikel lesen | |
| NOVO NORDISK | 37,705 | -1,17 % | Novo Nordisk-Aktie: Droht jetzt ein Kurssturz um -30%? | Der jüngste Aufwärtstrend der Novo Nordisk-Aktie ging Mitte Mai zu Ende. Der Kurs des dänischen Pharmakonzerns schaffte es nicht mehr über die Hürde von 300 DKK. Und nun droht nach Einschätzung eines... ► Artikel lesen | |
| PFIZER | 22,020 | -0,07 % | BARCLAYS stuft PFIZER INC auf 'Underweight' | LONDON (dpa-AFX Analyser) - Die britische Investmentbank Barclays hat die Einstufung für Pfizer mit einem Kursziel von 25 US-Dollar auf "Underweight" belassen. Angesichts der bisherigen Erfahrungen... ► Artikel lesen | |
| ELI LILLY | 929,40 | -0,10 % | HBM Healthcare Investments AG: Eli Lilly übernimmt HBM-Portfoliounternehmen Curevo für bis zu USD 1.5 Milliarden | HBM Healthcare Investments AG
/ Schlagwort(e): Ankauf
Eli Lilly übernimmt HBM-Portfoliounternehmen Curevo für bis zu USD 1.5 Milliarden
26.05.2026 / 13:10 CET/CEST
Die... ► Artikel lesen | |
| BRIGHT MINDS BIOSCIENCES | 81,47 | +0,56 % | Bright Minds Biosciences Announces Closing of US$175 Million Public Offering | NEW YORK, Jan. 09, 2026 (GLOBE NEWSWIRE) -- Bright Minds Biosciences Inc. (CSE: DRUG) (NASDAQ: DRUG) ("Bright Minds" or the "Company") today closed its recently announced public offering of 1,945... ► Artikel lesen | |
| ETON PHARMACEUTICALS | 28,680 | 0,00 % | Eton Pharmaceuticals Expands Rare Disease Portfolio Through Agreement for U.S. Commercialization Rights to IMPAVIDO (miltefosine) | IMPAVIDO is the first and only FDA-approved oral therapy for visceral, cutaneous, and mucosal leishmaniasis caused by specific Leishmania speciesLeishmaniasis is a rare but potentially life-threatening... ► Artikel lesen | |
| VERTEX PHARMACEUTICALS | 375,95 | -0,27 % | Vertex: FDA Accepts BLA Submission For Povetacicept; Assigns PDUFA Target Action Date Of Nov. 30 | CAMBRIDGE (MASSACHUSETTS) (dpa-AFX) - Vertex Pharmaceuticals Incorporated (VRTX) announced the FDA has accepted its Biologics License Application submission for povetacicept, an investigational... ► Artikel lesen | |
| GENERATE BIOMEDICINES | 12,970 | 0,00 % | Generate:Biomedicines: Generate Biomedicines, Inc. Reports First Quarter 2026 Financial Results and Provides Business Update | SOLAIRIA-1 AND SOLAIRIA-2 Phase 3 clinical trials for GB-0895 (anti-TSLP) continue to advance
Clinical trial sites activated for GB-4362 (MMAE neutralizer); first... ► Artikel lesen | |
| NOVARTIS | 125,94 | +0,27 % | DEUTSCHE BANK RESEARCH stuft NOVARTIS AG auf 'Buy' | FRANKFURT (dpa-AFX Analyser) - Deutsche Bank Research hat die Einstufung für Novartis anlässlich der Investorenveranstaltung "dbAccess European Champions Conference" mit einem Kursziel von 135 Franken... ► Artikel lesen | |
| ABBOTT LABORATORIES | 75,46 | +0,05 % | EQS-News: Abbott erhält CE-Kennzeichnung für die weltweit erste duale Glukose-Keton-Sensortechnologie für Menschen mit Diabetes | EQS-News: Abbott
/ Schlagwort(e): Expansion/Sonstiges
Abbott erhält CE-Kennzeichnung für die weltweit erste duale Glukose-Keton-Sensortechnologie für Menschen mit Diabetes
27.05.2026... ► Artikel lesen |
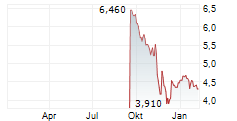
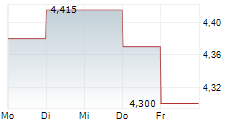
Das Unternehmen INFANT BACTERIAL THERAPEUTICS AB kann der Branche Pharma zugeordnet werden. Der aktuelle Kurs liegt bei 5,160 Euro und damit +0,78 % im Plus.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu INFANT BACTERIAL THERAPEUTICS AB finden Sie auf Wallstreet Online.