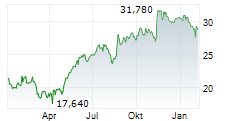
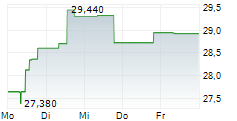
| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
| 34,920 | 35,000 | 14:52 | |
| 34,720 | 35,200 | 01.06. |
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| Mo | ALK Abello: Report on transactions with ALK-Abelló A/S B shares and associated securities by managerial staff | 2 | GlobeNewswire (USA) | ||
| 05.05. | ALK-Abelló A/S reports Q1 results, raises guidance | 2 | Seeking Alpha | ||
| 05.05. | ALK-Abelló Profit Advances In Q1 | 2 | RTTNews | ||
| 04.05. | ALK Abello: Three-month interim report (Q1) 2026 (unaudited) | 370 | GlobeNewswire (Europe) | ALK delivers 18% organic revenue growth with operating profit (EBIT) up 22% in Q1 Q1 saw broad-based growth across key categories and regions with sustained, strong momentum in tablet sales in Europe... ► Artikel lesen | |
| 04.05. | ALK Abello: ALK upgrades its full-year outlook | 269 | GlobeNewswire (Europe) | Inside Information ALK (ALKB:DC / Nasdaq Copenhagen: ALK B) today announced that the 2026 full-year financial outlook has been upgraded reflecting a strong underlying momentum for tablet sales and... ► Artikel lesen | |
| 28.04. | ALK Abello: Invitation to the presentation of ALK's first quarter 2026 results on Tuesday, 5 May 2026 | 8 | GlobeNewswire (USA) | ||
| ALK-ABELLO Aktie jetzt für 0€ handeln | |||||
| 20.04. | ALK Abello: ALK announces successful outcome of phase 2 trial with its tablet for treatment of peanut allergy demonstrating early onset of efficacy | 658 | GlobeNewswire (Europe) | Inside Information The ALLIANCE phase 2 trial demonstrated proof-of-concept after only 6 months of maintenance treatment in a broad patient population comprising both children and adults.Clear dose-dependent... ► Artikel lesen | |
| 15.04. | ALK Abello: neffy approved as the first needle-free anaphylaxis treatment of adults and children in Canada | 221 | GlobeNewswire (Europe) | ALK (ALKB:DC / Nasdaq Copenhagen: ALK B) today announced that Health Canada has approved neffy 2 mg for the emergency treatment of allergic reactions (anaphylaxis) due to insect stings or bites, foods... ► Artikel lesen | |
| 08.04. | ALK Abello: Report on transactions with ALK-Abelló A/S B shares and associated securities by managerial staff | 2 | GlobeNewswire (USA) | ||
| 01.04. | ALK Abello: Grant of share-based long-term incentive instruments to members of the Board of Management and key employees | 3 | GlobeNewswire (USA) | ||
| 31.03. | ALK Abello: EURneffy 1 mg approved across the EU as the first and only needle-free adrenaline treatment for young children (=15 kg) at risk of anaphylaxis | 427 | GlobeNewswire (Europe) | Today, ALK announced that children aged 4 and older living with severe allergies across the EU can now access the first and only needle-free adrenaline treatment for anaphylaxis, after the European... ► Artikel lesen | |
| 17.03. | Dividendenbekanntmachungen (17.03.2026) | 9.441 | wallstreetONLINE (Dividenden) | Unternehmen ISIN-Code Dividende (Währung) Dividende (EUR) 2020 BULKERS LTD BMG9156K1018 0,9713 NOK 0,0872 EUR ALK-ABELLO A/S DK0061802139 1,6 DKK 0,2141 EUR ARES MANAGEMENT CORPORATION US03990B1017 1... ► Artikel lesen | |
| 16.03. | ALK Abello: Annual General Meeting in ALK-Abelló A/S held on 16 March 2026 | 327 | GlobeNewswire (Europe) | ALK (ALKB:DC / Nasdaq Copenhagen: ALK B): ALK-Abelló A/S held its Annual General Meeting on 16 March 2026. The Annual General Meeting transacted the following business: Approval of the annual report... ► Artikel lesen | |
| 04.03. | ALK Abello: New data reveals 9 in 10 people prefer EURneffy, a needle-free nasal adrenaline spray, over auto-injectors | 790 | GlobeNewswire (Europe) | ALK presents new findings that directly compare user preference between needle-free nasal and injectable adrenaline treatments for anaphylaxis, demonstrating that 88% of participants prefer EURneffy-the... ► Artikel lesen | |
| 20.02. | ALK-Abelló A/S GAAP EPS of $5.40, revenue of $6.31B | 4 | Seeking Alpha | ||
| 20.02. | ALK Abello: Annual General Meeting in ALK-Abelló A/S on 16 March 2026 | 3 | GlobeNewswire (USA) | ||
| 20.02. | ALK-Abelló übertrifft Erwartungen im vierten Quartal und hebt Dividende an | 10 | Investing.com Deutsch | ||
| 20.02. | ALK Abello: Henriette Mersebach to step down as ALK's head of R&D | 230 | GlobeNewswire (Europe) | ALK (ALKB:DC / Nasdaq Copenhagen: ALK B) today announced that Henriette Mersebach will step down as Executive Vice President, Research and Development, and as a member of the Board of Management,... ► Artikel lesen | |
| 20.02. | ALK Abello: Annual report 2025: ALK delivers 15% revenue growth and 26% EBIT margin | 878 | GlobeNewswire (Europe) | ALK's (ALKB:DC / Nasdaq Copenhagen: ALK B) full-year results came in at the top end of the latest outlook, supported by solid performance in Q4 and continued commercial momentum. ALK expects sustained... ► Artikel lesen | |
| 13.02. | ALK Abello: Invitation to the presentation of ALK's annual report 2025 on Friday, 20 February 2026 | 2 | GlobeNewswire (USA) |
1 2 Weiter >>
33 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BAYER | 33,470 | -4,73 % | Märkte am Morgen: Delivery Hero, MTU, Lufthansa, TUI, Airbus, Bayer, Space X | Nach der außergewöhnlichen Feiertagskonstellation - Pfingstmontag mit Handel in Deutschland, Memorial Day ohne Handel in den USA - tritt der DAX heute leicht auf die Bremse (-0,3 % erwartet). Gestern... ► Artikel lesen | |
| NOVO NORDISK | 37,750 | -1,05 % | Novo Nordisk-Aktie: Droht jetzt ein Kurssturz um -30%? | Der jüngste Aufwärtstrend der Novo Nordisk-Aktie ging Mitte Mai zu Ende. Der Kurs des dänischen Pharmakonzerns schaffte es nicht mehr über die Hürde von 300 DKK. Und nun droht nach Einschätzung eines... ► Artikel lesen | |
| PFIZER | 22,020 | -0,07 % | BARCLAYS stuft PFIZER INC auf 'Underweight' | LONDON (dpa-AFX Analyser) - Die britische Investmentbank Barclays hat die Einstufung für Pfizer mit einem Kursziel von 25 US-Dollar auf "Underweight" belassen. Angesichts der bisherigen Erfahrungen... ► Artikel lesen | |
| NOVARTIS | 125,94 | +0,27 % | DEUTSCHE BANK RESEARCH stuft NOVARTIS AG auf 'Buy' | FRANKFURT (dpa-AFX Analyser) - Deutsche Bank Research hat die Einstufung für Novartis anlässlich der Investorenveranstaltung "dbAccess European Champions Conference" mit einem Kursziel von 135 Franken... ► Artikel lesen | |
| MERCK KGAA | 130,80 | 0,00 % | DAX-Check LIVE: Delivery Hero, Infineon, Merck, Nordex, Secunet, Siemens im Fokus | Der deutsche Aktienmarkt hat den Pfingstmontag mit einem fulminanten Kurssprung begangen. Der DAX startete mit Gewinnen, übersprang direkt die wichtige Marke von 25.000 Punkten und kletterte im Verlauf... ► Artikel lesen | |
| GILEAD SCIENCES | 112,40 | -0,27 % | Während Merck, Amgen, Novo Nordisk und Fresenius Milliarden bewegen, wächst die Fantasie im Zelltherapie-Sektor | ||
| AURORA CANNABIS | 3,144 | +0,58 % | War das erst der Anfang? Warum der Aurora-Deal und ein 50-Millionen-AUD-Vertrag Bioxyne weiteren Schub geben könnten | ||
| SANOFI | 73,88 | -0,40 % | DEUTSCHE BANK RESEARCH stuft SANOFI auf 'Buy' | FRANKFURT (dpa-AFX Analyser) - Deutsche Bank Research hat die Einstufung für Sanofi mit einem Kursziel von 100 Euro auf "Buy" belassen. In den Gesprächen mit den Franzosen sei es vor allem um den Umbruch... ► Artikel lesen | |
| GSK | 21,170 | -0,38 % | GSK erzielt teilweisen Therapieerfolg mit Hepatitis-B-Wirkstoff | DJ GSK erzielt teilweisen Therapieerfolg mit Hepatitis-B-Wirkstoff
Von Adria Calatayud
DOW JONES--GSK hat mit einem Medikamentenkandidaten gegen Hepatitis B einen teilweisen Therapieerfolg... ► Artikel lesen | |
| ABBVIE | 183,30 | +0,11 % | AbbVie Announces European Commission Approval of AQUIPTA (atogepant) for the Acute Treatment of Migraine in Adults | AQUIPTA (atogepant) is now approved in the European Union for the acute treatment of migraine in adults, with or without aura, and for the prophylaxis of migraine in adults who have at least... ► Artikel lesen | |
| ROCHE | 347,70 | -0,18 % | BARCLAYS stuft ROCHE HOLDINGS AG auf 'Overweight' | LONDON (dpa-AFX Analyser) - Die britische Investmentbank Barclays hat die Einstufung für Roche mit einem Kursziel von 410 Franken auf "Overweight" belassen. Analyst James Gordon berichtete am Montagabend... ► Artikel lesen | |
| CANOPY GROWTH | 0,935 | +0,45 % | Canopy Growth Corporation: Canopy Growth Relaunches Tweed Brand in Germany with New MTL Cannabis Strain Lineup, Marking First International Release following Acquisition | Pablo's Revenge, Dante'z Inferno, and Frost'd Flakes launched under Tweed brand Up to five additional strains expected to follow in June 2026 Company targeting rapid growth in Germany's medical... ► Artikel lesen | |
| STADA ARZNEIMITTEL | - | - | Enzymatica AB: Quarterly report Q1/2026 - Enzymatica strengthens international position through STADA agreement | First quarter
Net sales amounted to SEK 11.4 million (12.3).
Operating result amounted to SEK -13.6.million (-18.0)
Earnings per share, before... ► Artikel lesen | |
| ELI LILLY | 929,40 | -0,10 % | HBM Healthcare Investments AG: Eli Lilly übernimmt HBM-Portfoliounternehmen Curevo für bis zu USD 1.5 Milliarden | HBM Healthcare Investments AG
/ Schlagwort(e): Ankauf
Eli Lilly übernimmt HBM-Portfoliounternehmen Curevo für bis zu USD 1.5 Milliarden
26.05.2026 / 13:10 CET/CEST
Die... ► Artikel lesen | |
| MERCK & CO | 98,68 | -0,35 % | Ihre wichtigsten Termine: Alle Analysten-Augen auf: Aroundtown, Kingfisher sowie Merck & Co | © Foto: © 2023 Merck & CoGut geplant in den Tag. Mit dem wO-Tagesausblick haben Sie die wichtigsten Termine im Blick und starten bestens vorbereitet in den neuen Handelstag.Unternehmenstermine 06:45... ► Artikel lesen |
ALK-ABELLO A/S gehört der Branche Pharma an. Die relative Kursveränderung beträgt aktuell +0,51 % bei einem Aktienkurs von 35,140 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu ALK-ABELLO A/S finden Sie auf Wallstreet Online.