| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
| 2,840 | 2,885 | 18:55 |
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 26.05. | MaaT Pharma Provides Business Update and Reports Financial Results for the First Quarter 2026 | 568 | Business Wire | MaaT013 (Xervyteg):
In May 2026, the Company was informed of a "negative trend" opinion on its Marketing Authorization Application from the Committee for Medicinal Products for Human Use... ► Artikel lesen | |
| MAAT PHARMA Aktie jetzt für 0€ handeln | |||||
| 20.05. | MaaT Pharma Provides an Update on the Application for Marketing Authorization of MaaT013 (Xervyteg) in the treatment of acute Graft-versus-Host Disease | 632 | Business Wire | During the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) Oral Explanation, the Company received a "negative trend" opinion for the upcoming June... ► Artikel lesen | |
| 11.05. | MaaT Pharma to Hold Annual General Meeting on June 16, 2026 | 269 | Business Wire | Regulatory News:
MaaT Pharma (EURONEXT: MAAT the "Company"), a clinical-stage biotechnology company and a leader in the development of Microbiome Ecosystem TherapiesTM (MET) dedicated to enhancing... ► Artikel lesen | |
| 21.04. | Original-Research: MaaT Pharma SACA (von First Berlin Equity Research GmbH): Buy | 285 | EQS Group (EN) | Original-Research: MaaT Pharma SACA - from First Berlin Equity Research GmbH
21.04.2026 / 15:09 CET/CEST
Dissemination of a Research, transmitted by EQS News - a service of EQS Group.
The issuer... ► Artikel lesen | |
| 01.04. | MaaT Pharma Announces Publication of Retrospective Data in Third-Line Acute GvHD from the CHRONOS Study in Bone Marrow Transplantation Journal | 397 | Business Wire | CHRONOS is one of the largest real-world studies including refractory GI-aGvHD patients (n=59) treated with third-line best available treatments other than microbiome-based therapy
Results... ► Artikel lesen | |
| 31.03. | MaaT Pharma Announces 2025 Annual Results and Provides Business Updates | 471 | Business Wire | Potential Pivotal Year for MaaT013 (Xervyteg):
Marketing Authorization Application (MAA) in Europe for MaaT013 (Xervyteg) submitted to the European Medicines Agency (EMA) in June 2025
... ► Artikel lesen | |
| 23.03. | MaaT Pharma presents phase 3 trial results for aGvHD treatment | 2 | Investing.com | ||
| 23.03. | MaaT Pharma Presents the ARES Phase 3 Pivotal Trial Final Data During the Presidential Plenary Session at the 52nd Annual Meeting of the European Society for Bone and Marrow Transplantation | 363 | Business Wire | Regulatory News:
MaaT Pharma (EURONEXT: MAAT the "Company"), a clinical-stage biotechnology company and a leader in the development of Microbiome Ecosystem TherapiesTM (MET) dedicated to enhancing... ► Artikel lesen | |
| 09.03. | MaaT Pharma to Present Four Abstracts at the 52nd European Bone Marrow Transplantation Annual Meeting and to Highlight Clinigen-Hosted Industry Symposium on acute GvHD Management | 567 | Business Wire | Oral presentation during Presidential Plenary Session to feature the final results of the ARES pivotal Phase 3 trial evaluating MaaT013 in acute Graft-versus-Host Disease (aGvHD) with gastrointestinal... ► Artikel lesen | |
| 03.02. | MaaT Pharma: Monthly Information Regarding the Total Number of Voting Rights and Shares Comprising the Share Capital | 392 | Business Wire | Article 223-16 of the General Regulations of the Financial Markets Authority
(AMF Autorité des Marchés Financiers)
Regulatory News:
MaaT Pharma (EURONEXT: MAAT the "Company"), a clinical-stage... ► Artikel lesen | |
| 20.01. | MaaT Pharma startet Phase-2-Studie mit Mikrobiom-Therapie bei Lungenkrebs | 9 | Investing.com Deutsch | ||
| 20.01. | MaaT Pharma Announces First Patient Randomized in IMMUNOLIFE Phase 2 Study Sponsored by Gustave Roussy, To Explore the Role of the Gut Microbiome To Overcome ICI Resistance in Advanced NSCLC Patients with Antibiotic-Induced Dysbiosis | 646 | Business Wire | IMMUNOLIFE, a Phase 2 randomized exploratory study, will evaluate the potential of MaaT033 in combination with Cemiplimab versus Best Investigator Choice (i.e.: second line) in enhancing disease... ► Artikel lesen | |
| 10.12.25 | Original-Research: MaaT Pharma SACA (von First Berlin Equity Research GmbH): BUY | 459 | EQS Group (EN) | Original-Research: MaaT Pharma SACA - from First Berlin Equity Research GmbH
10.12.2025 / 11:25 CET/CEST
Dissemination of a Research, transmitted by EQS News - a service of EQS Group.
The issuer... ► Artikel lesen | |
| 08.12.25 | MaaT Pharma reports 54% one-year survival rate in pivotal GvHD trial | 4 | Investing.com | ||
| 08.12.25 | MaaT Pharma meldet 54 % Ein-Jahres-Überlebensrate in zulassungsrelevanter GvHD-Studie | 6 | Investing.com Deutsch | ||
| 08.12.25 | MaaT Pharma Presents Pivotal ARES Phase 3 Results for MaaT013 (Xervyteg) in Acute GvHD at ASH 2025 Annual Congress and Announces 54% 1-Year Overall Survival | 778 | Business Wire | Presentation included previously disclosed primary results from the pivotal ARES Phase 3 single-arm trial evaluating MaaT013 (Xervyteg) in treating refractory severe acute Graft-versus-Host Disease... ► Artikel lesen | |
| 19.11.25 | Original-Research: MaaT Pharma SACA (von First Berlin Equity Research GmbH): BUY | 419 | EQS Group (EN) | Original-Research: MaaT Pharma SACA - from First Berlin Equity Research GmbH
19.11.2025 / 11:11 CET/CEST
Dissemination of a Research, transmitted by EQS News - a service of EQS Group.
The issuer... ► Artikel lesen | |
| 17.11.25 | XFRA 4RD: WIEDERAUFNAHME/RESUMPTION | 237 | Xetra Newsboard | FOLGENDE(S) INSTRUMENT(E) WIRD/ WERDEN WIEDER IN DEN HANDEL AUFGENOMMEN MIT FOLGENDEM TRADING SCHEDULE.THE FOLLOWING INSTRUMENT(S) IS/ARE RESUMED TRADING WITH FOLLOWING TRADING SCHEDULE:INSTRUMENT NAME... ► Artikel lesen | |
| 14.11.25 | MaaT Pharma Announces the Successful Completion of Its Global Offering of €9.1 Million | 554 | Business Wire | Regulatory News:
THIS ANNOUNCEMENT AND THE INFORMATION CONTAINED HEREIN IS RESTRICTED AND IS NOT FOR RELEASE, PUBLICATION OR DISTRIBUTION, IN WHOLE OR IN PART, DIRECTLY OR INDIRECTLY, IN, INTO... ► Artikel lesen | |
| 14.11.25 | XFRA 4RD: AUSSETZUNG/SUSPENSION | 376 | Xetra Newsboard | DAS/ DIE FOLGENDE(N) INSTRUMENT(E) IST/ SIND AB SOFORT AUSGESETZT:THE FOLLOWING INSTRUMENT(S) IS/ ARE SUSPENDED WITH IMMEDIATE EFFECT:INSTRUMENT NAME KUERZEL/SHORTCODE ISIN BIS/UNTILMAAT PHARMA S.A.... ► Artikel lesen |
1 2 Weiter >>
34 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BIONTECH | 76,30 | -3,72 % | Vorsicht bei BioNTech! Novo Nordisk gibt Gas! Kaufchance bei Vidac Pharma Aktie! | Kaufstimmung bei Novo Nordisk. Die Aktie des dänischen Pharmagiganten scheint den seit Sommer 2024 begonnenen Abwärtstrend zu verlassen. Anleger reagieren auf positive Meldungen. Bei Vidac Pharma ist... ► Artikel lesen | |
| BB BIOTECH | 46,150 | -2,02 % | BB Biotech: "Wachstumsstarke Mid- und Small Caps bilden das Rückgrat unseres Portfolios": Mehr Einzelwerte und ... | ||
| BRAIN BIOTECH | 2,910 | -3,00 % | EQS-News: BRAIN Biotech AG: BRAIN Biotech veröffentlicht Halbjahreszahlen für das Geschäftsjahr 2025/26 und bestätigt Gesamtjahresprognose | EQS-News: BRAIN Biotech AG
/ Schlagwort(e): Halbjahresergebnis/Halbjahresbericht
BRAIN Biotech veröffentlicht Halbjahreszahlen für das Geschäftsjahr 2025/26 und bestätigt Gesamtjahresprognose... ► Artikel lesen | |
| OCUGEN | 1,152 | -1,87 % | Canaccord lowers Ocugen stock price target to $11 on model update | ||
| VIKING THERAPEUTICS | 25,500 | -5,20 % | Viking Therapeutics' Q3 Catalyst That Could Turn Eli Lilly's and Novo Nordisk's Daily Weight Loss Pill Strategy Upside Down | ||
| INFLARX | 1,930 | -2,03 % | InflaRx NV unter massivem Druck: Was die wenigsten Anleger auf dem Schirm haben - Was ich konkret unternommen habe! | ||
| INTELLIA THERAPEUTICS | 11,800 | -0,17 % | Intellia to present Phase 3 gene editing trial data at congress | ||
| EDITAS MEDICINE | 2,790 | -2,45 % | Editas Medicine prices $125M stock offering with warrants | ||
| KUROS BIOSCIENCES | 20,500 | -1,25 % | EILMELDUNG bei Kuros Biosciences: Was sich hinter den Kulissen tut - Meine Analyse im Detail | ||
| SAREPTA THERAPEUTICS | 14,700 | -2,91 % | Biotech-Player: Zelltherapie als Comeback-Story im Milliardenmarkt. Neue Ideen nach Corona von BioNTech, Qiagen und Biotech AG | ||
| PACIFIC BIOSCIENCES OF CALIFORNIA | 1,340 | -1,72 % | Druck auf Pacific Biosciences nimmt zu: Warum die Geduld der Anleger auf eine harte Probe gestellt wird | Pacific Biosciences gerät zunehmend unter Druck: Die Aktie steht nach einer "significant downward revision of revenue guidance" und dem erzwungenen Aktienrückzug eines Großaktionärs im Fokus. Für Investoren... ► Artikel lesen | |
| CARDIOL THERAPEUTICS | 1,036 | -1,33 % | Cardiol Therapeutics Inc. - 6-K, Report of foreign issuer | ||
| BASILEA | 53,90 | -2,00 % | Basilea Pharmaceutica International Ltd, Allschwil: Basilea erhält weitere USD 13 Mio. von BARDA zur Entwicklung des neuartigen Antibiotikums Ceftibuten-Ledaborbactam | Ad hoc-Mitteilung gemäss Art. 53 KR
Allschwil, 28. Mai 2026
Basilea Pharmaceutica AG, Allschwil (SIX: BSLN), ein biopharmazeutisches Unternehmen mit bereits vermarkteten Produkten und dem Ziel... ► Artikel lesen | |
| ARROWHEAD PHARMACEUTICALS | 66,40 | +2,75 % | Arrowhead Pharmaceuticals, Inc.: Arrowhead Pharmaceuticals Presents New Positive Clinical Cardiometabolic Data at the 94th European Atherosclerosis Society (EAS) Congress | New data support potential use of plozasiran without dose adjustment in patients with moderate-to-severe renal or moderate hepatic impairment
Case report suggests that preconception exposure to... ► Artikel lesen | |
| MICROBOT MEDICAL | 1,710 | +0,59 % | Microbot Medical Inc. - 8-K, Current Report |
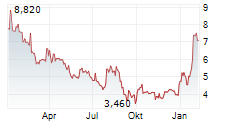
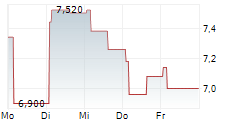
MAAT PHARMA SA gehört der Branche Biotechnologie an. Der aktuelle Kurs liegt bei 2,870 Euro und damit +0,35 % im Plus.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu MAAT PHARMA SA finden Sie auf Wallstreet Online.