| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
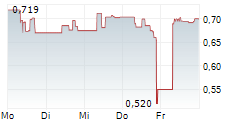
| 0,565 | 0,583 | 22.05. | |
| 0,564 | 0,586 | 22.05. |
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| Fr | SBP GROUP (01177): VOLUNTARY ANNOUNCEMENT - DATA FROM TWO CLINICAL STUDIES OF M701 "CD3/EPCAM BISPECIFIC ANTIBODY" PRESENTED AT 2026 ASCO | 4 | HKEx | ||
| Do | SBP GROUP (01177): VOLUNTARY ANNOUNCEMENT - ZONGERTINIB, A HER2 TYROSINE KINASE INHIBITOR, APPROVED FOR FIRST-LINE TREATMENT OF HER2-MUTANT NON-SMALL ... | 8 | HKEx | ||
| SINO BIOPHARM Aktie jetzt für 0€ handeln | |||||
| 15.05. | SBP GROUP (01177): NEXT DAY DISCLOSURE RETURN | 1 | HKEx | ||
| 14.05. | SBP GROUP Announces Preclinical Data of Innovative Drug TQF6422 at European Congress on Obesity | 5 | AASTOCKS | ||
| 14.05. | SBP GROUP (01177): VOLUNTARY ANNOUNCEMENT - LATEST RESEARCH DATA ON TQF6422 "ACTRII A/B MONOCLONAL ANTIBODY" PRESENTED AT ECO 2026 | 3 | HKEx | ||
| 12.05. | GSK and SBP Group team up to advance bepirovirsen launch in China | 3 | Pharmaceutical Technology | ||
| 12.05. | G Sachs Raises SBP GROUP TP to HKD8.05, Maintains Buy on Introduction of GSK Hepatitis B Drug | 6 | AASTOCKS | ||
| 11.05. | GSK strikes agreement with China's SBP Group, launches final tranche of buyback programme | 4 | Sharecast | ||
| 11.05. | SBP GROUP Forms Exclusive Strategic Partnership with GlaxoSmithKline to Accelerate Mainland Launch of bepirovirsen | 4 | AASTOCKS | ||
| 11.05. | SBP GROUP (01177): VOLUNTARY ANNOUNCEMENT - ENTERING INTO AN EXCLUSIVE STRATEGIC COLLABORATION WITH GSK | 4 | HKEx | ||
| 07.05. | SBP GROUP (01177): NEXT DAY DISCLOSURE RETURN | 1 | HKEx | ||
| 07.05. | SBP GROUP's Category 1 Innovative Drug M701 NDA Accepted | 3 | AASTOCKS | ||
| 07.05. | SBP GROUP (01177): VOLUNTARY ANNOUNCEMENT - ACCEPTANCE OF NEW DRUG APPLICATION FOR M701 "CD3/EPCAM BISPECIFIC ANTIBODY" | 2 | HKEx | ||
| 07.05. | SBP GROUP CCR8 Monoclonal Antibody for Gastric Cancer Completes First Patient Enrollment in Clinical Trial | 4 | AASTOCKS | ||
| 07.05. | SBP GROUP (01177): VOLUNTARY ANNOUNCEMENT - FIRST PATIENT ENROLLMENT IN THE PHASE III REGISTRATIONAL CLINICAL TRIAL OF CAFELKIBART "CCR8 MONOCLONAL ANTIBODY" ... | 2 | HKEx | ||
| 06.05. | SBP GROUP: Kumoxili Capsules Approved in Mainland for 2nd Indication | 7 | AASTOCKS | ||
| 06.05. | SBP GROUP (01177): VOLUNTARY ANNOUNCEMENT - NEW INDICATION OF CULMERCICLIB CAPSULE "CDK2/4/6 INHIBITOR" FOR FIRST-LINE TREATMENT OF BREAST CANCER APPROVED ... | 2 | HKEx | ||
| 30.04. | SBP GROUP (01177): NEXT DAY DISCLOSURE RETURN | 2 | HKEx | ||
| 29.04. | SBP GROUP (01177): 2025 ENVIRONMENTAL, SOCIAL AND GOVERNANCE REPORT | 4 | HKEx | ||
| 29.04. | SBP GROUP (01177): NOTIFICATION LETTER AND REQUEST FORM FOR NON-REGISTERED SHAREHOLDERS | 3 | HKEx |
1 2 3 4 5 Weiter >>
82 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BIONTECH | 79,35 | +1,67 % | Aktionärinnen und Aktionäre der BioNTech SE stimmen auf der ordentlichen Hauptversammlung 2026 allen Tagesordnungspunkten zu | MAINZ, Deutschland, 15. Mai 2026 (GLOBE NEWSWIRE) -- BioNTech SE (Nasdaq: BNTX, "BioNTech" oder "das Unternehmen") hat heute die diesjährige ordentliche Hauptversammlung abgehalten. Insgesamt waren... ► Artikel lesen | |
| EVOTEC | 5,230 | +2,95 % | KAUFKURSE bei Renk und Desert Gold! SCHOCK bei der Evotec Aktie! | Während der Goldpreis schwächelt, befindet sich die Aktie von Desert Gold in einem sauberen Aufwärtstrend. Und geht es nach Analysten, ist eine Vervielfachung möglich. Denn Desert Gold wird in wenigen... ► Artikel lesen | |
| BB BIOTECH | 48,100 | +0,42 % | Aktien KW 17 Märkte im Würgegriff. Planloser Trump , selbstbewusterer Iran. Lösungen müssen her. News. Rheinmetall. ITM Power. Porsche. The Platform Group. K+S. Bugatti. Circus. Surteco. NanoRepro. BB Biotech. Mensch und Maschine. ATOSS. MHP Hotel | Aktien - letzte Woche warteten die Märkte auf eine Lösung des Iran-Kriegs. Aber die Iraner sehen sich im Vorteil und Trump verlängert "auf unbestimmte Zeit" den Waffenstillstand. Aber die Strasse von... ► Artikel lesen | |
| MEDIGENE | 0,037 | +18,59 % | PTA-Adhoc: Medigene AG: Widerruf der Zulassung der Aktien der Medigene AG zum Handel im regulierten Markt | DJ PTA-Adhoc: Medigene AG: Widerruf der Zulassung der Aktien der Medigene AG zum Handel im regulierten Markt
Veröffentlichung von Insiderinformationen gemäß Artikel 17 MAR Schlagwort(e): Sonstiges... ► Artikel lesen | |
| QIAGEN | 30,290 | +0,65 % | QIAGEN N.V.: QIAGEN to advance AI-driven drug discovery with graph-based AI and curated bioinformatics knowledge with NVIDIA | Collaboration integrates NVIDIA accelerated computing and the NVIDIA BioNeMo platform with QIAGEN Digital Insights' curated biomedical knowledge bases Goal to advance how disease mechanisms... ► Artikel lesen | |
| MODERNA | 40,375 | -0,05 % | Moderna Flu Shot Heads To FDA Advisory Panel | ||
| PAION | 0,072 | -25,52 % | XFRA MISTRADE ANTRAG IN ISIN DE000A3E5EG5 WURDE STATTGEGEBEN | Dem Mistrade-Antrag wurde stattgegeben. Folgende Geschaefte in der ISIN DE000A3E5EG5, Wertpapier-Name: PAION AG INH O.N., wurden aufgehoben:ISIN Datum Zeit Volumen PreisDE000A3E5EG5 02.02.2026 08:13:40 300 0... ► Artikel lesen | |
| VALNEVA | 2,490 | +0,81 % | Valneva Aktie: Prognose kassiert - die Börse schaut woanders hin | Valneva hat das erste Quartal 2026 mit einem deutlichen Umsatzrückgang abgeschlossen und die Jahresprognose gesenkt. Der Aktienkurs reagierte nicht entscheidend, denn der Markt richtet seinen Blick... ► Artikel lesen | |
| AMGEN | 292,50 | +0,05 % | Dividendenbekanntmachungen (15.05.2026) | Unternehmen ISIN-Code Dividende (Währung) Dividende (EUR) A10 NETWORKS INC US0021211018 0,06 USD 0,0514 EUR AF GRUPPEN ASA NO0003078107 6,5 NOK 0,6029 EUR AGCO CORPORATION US0010841023 0... ► Artikel lesen | |
| EPIGENOMICS | 0,870 | 0,00 % | PTA-HV: Epigenomics AG: Einladung zur ordentlichen Hauptversammlung | DJ PTA-HV: Epigenomics AG: Einladung zur ordentlichen Hauptversammlung
Hauptversammlung gemäß -- 121 Abs. 4a AktG
Epigenomics AG: Einladung zur ordentlichen Hauptversammlung
Heidelberg... ► Artikel lesen | |
| NOVAVAX | 7,930 | 0,00 % | Novavax, Inc.: Novavax Reports First Quarter 2026 Financial Results and Operational Highlights | Total revenue of $140 million in the first quarter of 2026
Pfizer agreement announced in January for non-exclusive license to utilize Matrix-M® in two infectious... ► Artikel lesen | |
| STRYKER | 272,50 | -0,07 % | Stryker Buys Amplitude Vascular Systems For Upto $835 Mln To Expand Vascular Treatment Business | WASHINGTON (dpa-AFX) - Tuesday, BioStar Capital announced that Amplitude Vascular Systems, Inc., a medical technology company, has been acquired by Stryker Corporation (SYK) at a purchase price... ► Artikel lesen | |
| BIOGEN | 166,64 | -0,22 % | XETR DELETION OF INSTRUMENTS FROM XETRA - 22.05.2026 | The following instruments on XETRA do have their last trading day on 22.05.2026.Die folgenden Instrumente auf XETRA haben ihren letzten Handelstag am 22.05.2026..ISIN Name US09062X1037 Biogen Inc.US2521311074 DexCom... ► Artikel lesen | |
| BIOFRONTERA | 2,390 | -1,65 % | Biofrontera stock rating cut to Speculative Buy by Benchmark | ||
| HEIDELBERG PHARMA | 2,740 | -0,72 % | EQS-HV: Heidelberg Pharma AG: Bekanntmachung der Einberufung zur Hauptversammlung am 23.06.2026 in Ladenburg mit dem Ziel der europaweiten Verbreitung gemäß §121 AktG | EQS-News: Heidelberg Pharma AG
/ Bekanntmachung der Einberufung zur Hauptversammlung
Heidelberg Pharma AG: Bekanntmachung der Einberufung zur Hauptversammlung am 23.06.2026 in... ► Artikel lesen |
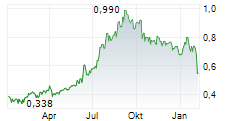
SINO BIOPHARMACEUTICAL LTD gehört der Branche Biotechnologie an. Mit einer aktuellen Kursveränderung von -0,010 (-1,70 %) liegt der Kurs bei 0,565 Euro. Beim derzeitigen Kurs ergibt sich, basierend auf einer gesamten Ausschüttung von 0,01 Euro in den vergangenen 12 Monaten, eine Dividendendrendite von +1,77 %.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu SINO BIOPHARMACEUTICAL LTD finden Sie auf Wallstreet Online.