- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| Do | Dermata Therapeutics, Inc. - S-8, Securities to be offered to employees in employee benefit plans | 4 | SEC Filings | ||
| 27.05. | Dermata Therapeutics, Inc. - 8-K, Current Report | - | SEC Filings | ||
| 13.05. | Dermata Therapeutics GAAP EPS of -$0.48 beats by $0.05 | 3 | Seeking Alpha | ||
| 13.05. | Dermata Therapeutics Provides Corporate Update and Reports Financial Results for the First Quarter 2026 | 274 | ACCESS Newswire | - Dermata announced their Tome Skincare brand for direct-to-consumer ("DTC") skincare products -- Dermata plans to launch its first DTC product, the Tome Foundational Treatment, a once-weekly mask to... ► Artikel lesen | |
| 13.05. | Dermata Therapeutics, Inc. - 10-Q, Quarterly Report | 2 | SEC Filings | ||
| 13.05. | Dermata Therapeutics, Inc. - 8-K, Current Report | 2 | SEC Filings | ||
| DERMATA THERAPEUTICS Aktie jetzt für 0€ handeln | |||||
| 23.04. | RBC Capital initiates Dermata Therapeutics stock at outperform | 2 | Investing.com | ||
| 02.04. | Dermata erhält australische Patentannahme für topischen Dermalfiller | 4 | Investing.com Deutsch | ||
| 02.04. | Dermata Therapeutics: Dermata Receives Notice of Acceptance of Australian Patent Application for Topical Application of Dermal Fillers with its Bioneedle Delivery System | 394 | ACCESS Newswire | - This would be Dermata's first patent, if issued, covering the use of its Bioneedle Delivery System ("BDS") with dermal fillers -- The Company also has allowed and issued patents in the US, Australia... ► Artikel lesen | |
| 27.03. | Dermata Therapeutics GAAP EPS of -$8.16 | 3 | Seeking Alpha | ||
| 26.03. | Dermata Therapeutics Provides Corporate Update and Reports Financial Results for the Year Ended 2025 | 1.966 | ACCESS Newswire | - Dermata announced a strategic pivot to develop and commercialize direct-to-consumer ("DTC") skincare products under the brand name Tome -- Dermata plans to launch its first DTC product, our Foundational... ► Artikel lesen | |
| 26.03. | Dermata Therapeutics, Inc. - 10-K, Annual Report | 4 | SEC Filings | ||
| 26.03. | Dermata Therapeutics, Inc. - 8-K, Current Report | 2 | SEC Filings | ||
| 10.03. | Dermata Therapeutics names Kyra Peckaitis VP of marketing | 2 | Investing.com | ||
| 10.03. | Dermata Therapeutics: Dermata Announces Appointment of Kyra Peckaitis as Vice President, Marketing | 2.512 | ACCESS Newswire | - Kyra Peckaitis will lead the launch of Dermata's recently announced Tome skincare brand - SAN DIEGO, CA / ACCESS Newswire / March 10, 2026 / Dermata Therapeutics, Inc. ("Dermata," or the "Company")... ► Artikel lesen | |
| 05.03. | Dermata Therapeutics: Dermata Opens a New Chapter with the Release of Tome Skincare | 718 | ACCESS Newswire | -The oldest new thing in skincare -- Dermata expects to launch its first product in the middle of 2026 - SAN DIEGO, CA / ACCESS Newswire / March 5, 2026 / Dermata Therapeutics, Inc. (Nasdaq:DRMA)(Nasdaq:DRMAW)... ► Artikel lesen | |
| 25.02. | Dermata Therapeutics, Inc. - 8-K, Current Report | 1 | SEC Filings | ||
| 03.02. | Dermata Therapeutics, Inc. - 8-K, Current Report | 1 | SEC Filings | ||
| 27.01. | Dermata Therapeutics, Inc. - 8-K, Current Report | 1 | SEC Filings | ||
| 20.01. | Dermata receives Australian patent for Spongilla acne treatment | 2 | Investing.com |
1 2 Weiter >>
35 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BIONTECH | 77,05 | -1,34 % | BioNTech und BMS melden neue Daten für Lungenkrebskandidat Pumitamig | BioNTech SE und Bristol Myers Squibb haben auf dem ASCO-Jahreskongress in Chicago erste globale Phase-2-Daten für den bispezifischen Immunmodulator Pumitamig in Kombination mit Chemotherapie bei fortgeschrittenem... ► Artikel lesen | |
| EVOTEC | 4,932 | -3,58 % | EQS-News: Evotec SE: Evotec ernennt Rui Wang zum Head of Global In Silico & AI | EQS-News: Evotec SE
/ Schlagwort(e): Sonstiges
Evotec ernennt Rui Wang zum Head of Global In Silico & AI
02.06.2026 / 07:30 CET/CEST
Für den Inhalt der Mitteilung ist... ► Artikel lesen | |
| BB BIOTECH | 45,850 | -0,86 % | BB Biotech: "Wachstumsstarke Mid- und Small Caps bilden das Rückgrat unseres Portfolios": Mehr Einzelwerte und ... | ||
| QIAGEN | 31,920 | -2,18 % | DEUTSCHE BANK RESEARCH stuft QIAGEN NV auf 'Buy' | FRANKFURT (dpa-AFX Analyser) - Deutsche Bank Research hat Qiagen mit einem Kursziel von 43 US-Dollar auf "Buy" belassen. Das Feedback aus Gesprächen mit etlichen US-Laborexperten deute darauf hin, dass... ► Artikel lesen | |
| MODERNA | 41,190 | +0,12 % | Opening Bell: Bitcoin, Strategy, Broadcom, Marvell, Micron, Axon Enterprise, Moderna, Lululemon | Die US-Börsen haben am Donnerstag eine zweigeteilte Entwicklung gezeigt. Der Dow Jones eröffnete mit klaren Gewinnen, baute diese im Verlauf deutlich aus und schloss 1,73 Prozent fester. Der Nasdaq... ► Artikel lesen | |
| PAION | 0,062 | -23,60 % | XFRA MISTRADE ANTRAG IN ISIN DE000A3E5EG5 WURDE STATTGEGEBEN | Dem Mistrade-Antrag wurde stattgegeben. Folgende Geschaefte in der ISIN DE000A3E5EG5, Wertpapier-Name: PAION AG INH O.N., wurden aufgehoben:ISIN Datum Zeit Volumen PreisDE000A3E5EG5 02.02.2026 08:13:40 300 0... ► Artikel lesen | |
| VALNEVA | 2,331 | -0,64 % | Valneva im Höhenflug: Das Signal das die meisten Anleger noch nicht kennen - Meine Einschätzung für die neue Börsenwoche | ||
| NOVAVAX | 8,080 | -1,70 % | NOVAVAX INC: Stabilität vor der nächsten Bewegung | ||
| STRYKER | 265,60 | +0,15 % | Leerink cuts Stryker stock price target to $407 on model update | ||
| BIOGEN | 171,56 | +1,19 % | Biogen's Salanersen Wins FDA Breakthrough Therapy Designation For Spinal Muscular Atrophy Treatment | WESTON (dpa-AFX) - Thursday, Biogen Inc. (BIIB) announced that the U.S. Food and Drug Administration has granted salanersen Breakthrough Therapy Designation for the treatment of spinal muscular... ► Artikel lesen | |
| BIOFRONTERA | 2,490 | -3,49 % | BIOFRONTERA AG zeigt bemerkenswerte Stärke | ||
| CRISPR THERAPEUTICS | 44,705 | -0,60 % | CRISPR Therapeutics AG (CRSP): One of The most Shorted Mid Cap Stocks to Buy on Robust Gene Editing Pipeline | ||
| REGENERON PHARMACEUTICALS | 551,40 | +0,04 % | CytomX Therapeutics Inc.: CytomX Announces Expansion of Strategic Research Collaboration with Regeneron in the Field of Conditional Bispecific Therapeutics for the Treatment of Cancer | - Expanded collaboration builds upon research momentum in developing next-generation bispecific immunotherapies using CytomX's PROBODY- and Regeneron's Veloci-Bi- platforms - - CytomX to receive... ► Artikel lesen | |
| BIOXXMED | 0,800 | -1,23 % | PTA-News: bioXXmed AG: Vollzug des Verkaufs der Rancoderm GmbH | DJ PTA-News: bioXXmed AG: Vollzug des Verkaufs der Rancoderm GmbH
Unternehmensmitteilung für den Kapitalmarkt
bioXXmed AG: Vollzug des Verkaufs der Rancoderm GmbH
Heidelberg (pta000/09.03.2026/13:50... ► Artikel lesen | |
| OCUGEN | 1,118 | +0,18 % | Ocugen, Inc. - 8-K, Current Report |
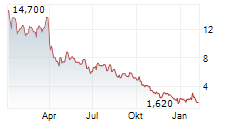
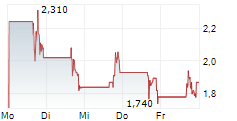
Das Unternehmen DERMATA THERAPEUTICS INC kann der Branche Biotechnologie zugeordnet werden. Der aktuelle Kurs liegt bei 1,190 Euro und damit +0,85 % im Plus.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu DERMATA THERAPEUTICS INC finden Sie auf Wallstreet Online.