| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
| 3,620 | 3,940 | 18:41 |
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| Di | Quoin Pharmaceuticals, Inc.: Quoin Pharmaceuticals Submits IND Application for QRX003 in Peeling Skin Syndrome | 109 | GlobeNewswire (Europe) | -First ever IND submission for this disease, which has no current treatment or cure- -Quoin plans Phase 2 study initiation in 2H 2026- -Submission supported by positive initial clinical data from... ► Artikel lesen | |
| 07.05. | Quoin Pharmaceuticals, Ltd. - 8-K, Current Report | - | SEC Filings | ||
| 07.05. | Quoin Pharmaceuticals, Ltd. - 10-Q, Quarterly Report | - | SEC Filings | ||
| 07.05. | Quoin Pharmaceuticals, Inc.: Quoin Pharmaceuticals Provides Corporate Update and Reports First Quarter 2026 Financial Results | 348 | GlobeNewswire (Europe) | - Filed Breakthrough Medicine Designation Application with Saudi FDA for QRX003 in Netherton Syndrome - Submitted Application to Japanese MHLW for Orphan Drug Designation (ODD) for QRX003; MHLW... ► Artikel lesen | |
| 30.04. | Quoin Pharmaceuticals, Inc.: Quoin Pharmaceuticals to Announce Corporate Update and First Quarter 2026 Financial Results on Thursday, May 7, 2026 | 1 | GlobeNewswire (USA) | ||
| 28.04. | Quoin plant neue klinische Studien für topisches Rapamycin-Präparat bei seltenen Erkrankungen | - | Investing.com Deutsch | ||
| QUOIN PHARMACEUTICALS Aktie jetzt für 0€ handeln | |||||
| 26.03. | Quoin Pharmaceuticals, Ltd. - 8-K, Current Report | 5 | SEC Filings | ||
| 26.03. | Quoin Pharmaceuticals, Inc.: Quoin Pharmaceuticals Provides Corporate Update and Reports Fourth Quarter and Full-Year 2025 Financial Results | 452 | GlobeNewswire (Europe) | - Closes Private Placement Financing of Up to $104.5 Million - Secures Orphan Drug Designations for QRX003 for Netherton Syndrome (NS) in the U.S. and Europe - Advances Proposed Expedited Regulatory... ► Artikel lesen | |
| 26.03. | FM Bank and Quoin Financial Bank migrate to Jack Henry | 5 | FinTech Futures | ||
| 25.03. | Quoin Pharmaceuticals Shares Rise 5% After Approval Path For QRX003 | 3 | RTTNews | ||
| 25.03. | Quoin Pharmaceuticals, Inc.: Quoin Pharmaceuticals Provides Clinical and Regulatory Update from Constructive Type C Meeting with U.S. FDA for QRX003 in Netherton Syndrome | 196 | GlobeNewswire (Europe) | FDA indicated that a single Phase 3 study may be sufficient to support marketing approval in the U.S. FDA expressed openness to an alternative study design for Phase 3 that would likely not include... ► Artikel lesen | |
| 19.03. | Quoin Pharmaceuticals, Inc.: Quoin Pharmaceuticals to Announce Corporate Update and Fourth Quarter and Full-Year 2025 Financial Results on Thursday, March 26, 2026 | 3 | GlobeNewswire (USA) | ||
| 11.03. | Quoin Pharmaceuticals, Inc.: Quoin Pharmaceuticals Announces FDA Grants Fast Track Designation for QRX003 for the Treatment of Netherton Syndrome | 265 | GlobeNewswire (Europe) | - Fast Track Designation facilitates development and expedites regulatory review of therapies addressing serious conditions with significant unmet medical need - - QRX003 lotion (4%) currently being... ► Artikel lesen | |
| 27.01. | Quoin Pharmaceuticals, Inc.: Quoin Pharmaceuticals Announces Submission to Japanese MHLW for Orphan Drug Designation for QRX003 in Netherton Syndrome | 191 | GlobeNewswire (Europe) | Confirmation Received from MHLW That QRX003 Qualifies for Both Orphan Drug Designation and Fast Track Regulatory Review Status Quoin Initiating the Establishment of a Japanese Subsidiary to Facilitate... ► Artikel lesen | |
| 20.01. | Quoin Pharmaceuticals, Ltd. - 8-K, Current Report | 1 | SEC Filings | ||
| 20.01. | Quoin Pharmaceuticals, Ltd.: Quoin Pharmaceuticals Files Breakthrough Medicine Designation Application in Saudi Arabia for QRX003 in Netherton Syndrome | 443 | GlobeNewswire (Europe) | If granted, QRX003 could be approved for sale and reimbursement in Saudi Arabia as a treatment for Netherton Syndrome in 2H 2026QRX003 could become the first ever approved treatment for Netherton Syndrome
ASHBURN... ► Artikel lesen | |
| 11.11.25 | Quoin Pharmaceuticals, Inc.: Quoin Pharmaceuticals Announces Achievement of Topical Rapamycin Target Loadings for Two Proprietary Delivery Technologies | 366 | GlobeNewswire (Europe) | Target Loadings of 4% and 5% Rapamycin Achieved for Topical Lotion and Dermal Patch Delivery Technologies Respectively Clinical Trial and Stability Batch Manufacture to Commence in Q4 2025 Clinical... ► Artikel lesen | |
| 06.11.25 | Quoin Pharmaceuticals, Inc.: Quoin Pharmaceuticals Announces Corporate Update and Third Quarter 2025 Financial Results | 262 | GlobeNewswire (Europe) | - Private Placement Financing of Up to $105.3 Million Closed in October 2025 to Fund Operations and R&D - Sally Lawlor Appointed CFO to Support Commercialization and Financial Operations - Orphan... ► Artikel lesen | |
| 04.11.25 | Prysmian Hosts Ribbon Cutting Ceremony to Unveil $63.8M Du Quoin Facility Expansion | 690 | ACCESS Newswire | DU QUOIN, IL / ACCESS Newswire / November 4, 2025 / Prysmian, a world leader in the energy transition and digital transformation, recently held a ribbon cutting ceremony to officially unveil the company's... ► Artikel lesen | |
| 21.10.25 | Quoin Pharmaceuticals, Inc.: Quoin Pharmaceuticals Announces U.S. FDA Grants Orphan Drug Designation for QRX003 in Netherton Syndrome | 658 | GlobeNewswire (Europe) | Upon Approval of NDA Quoin Will Receive Seven Years Marketing Exclusivity for QRX003 in the US Orphan Drug Designation previously granted by the European Medicines Agency in May 2025 ASHBURN, Va.... ► Artikel lesen |
1 2 Weiter >>
22 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BIONTECH | 76,20 | -0,13 % | Jetzt kommt die Neubewertung: UBS wettet auf BioNTechs Krebs-Showdown: 45 % Kurspotenzial! | © Foto: picture alliance / Laci Perenyi | Laci PerenyiDie Schweizer Großbank glaubt zunehmend an BioNTechs Krebsstrategie. Der ASCO-Kongress könnte jetzt zum großen Kurstreiber werden.BioNTech bekommt... ► Artikel lesen | |
| EVOTEC | 5,025 | -1,37 % | EQS-NVR: Evotec SE: Veröffentlichung der Gesamtzahl der Stimmrechte nach § 41 WpHG mit dem Ziel der europaweiten Verbreitung | EQS Gesamtstimmrechtsmitteilung: Evotec SE
/ Veröffentlichung der Gesamtzahl der Stimmrechte
Evotec SE: Veröffentlichung der Gesamtzahl der Stimmrechte nach § 41 WpHG mit dem... ► Artikel lesen | |
| BB BIOTECH | 45,600 | -1,19 % | BB Biotech: "Wachstumsstarke Mid- und Small Caps bilden das Rückgrat unseres Portfolios": Mehr Einzelwerte und ... | ||
| MEDIGENE | 0,020 | -31,97 % | PTA-Adhoc: Medigene AG: Widerruf der Zulassung der Aktien der Medigene AG zum Handel im regulierten Markt | DJ PTA-Adhoc: Medigene AG: Widerruf der Zulassung der Aktien der Medigene AG zum Handel im regulierten Markt
Veröffentlichung von Insiderinformationen gemäß Artikel 17 MAR Schlagwort(e): Sonstiges... ► Artikel lesen | |
| QIAGEN | 30,975 | +0,88 % | DEUTSCHE BANK RESEARCH stuft QIAGEN NV auf 'Buy' | FRANKFURT (dpa-AFX Analyser) - Deutsche Bank Research hat die Einstufung für Qiagen mit einem Kursziel von 43 US-Dollar auf "Buy" belassen. Das Interesse der Investoren habe vor allem dem Tuberkulosetest... ► Artikel lesen | |
| MODERNA | 40,585 | +3,47 % | Neue CAR-T-Erfolge bei Autoimmunerkrankungen: Warum Lösungen für Zelltherapien vor dem nächsten Wachstumsschub stehen könnten | ||
| PAION | 0,070 | -21,35 % | XFRA MISTRADE ANTRAG IN ISIN DE000A3E5EG5 WURDE STATTGEGEBEN | Dem Mistrade-Antrag wurde stattgegeben. Folgende Geschaefte in der ISIN DE000A3E5EG5, Wertpapier-Name: PAION AG INH O.N., wurden aufgehoben:ISIN Datum Zeit Volumen PreisDE000A3E5EG5 02.02.2026 08:13:40 300 0... ► Artikel lesen | |
| VALNEVA | 2,393 | -1,68 % | Original-Research: Valneva SE (von First Berlin Equity Research GmbH): Buy | Original-Research: Valneva SE - von First Berlin Equity Research GmbH
28.05.2026 / 17:08 CET/CEST
Veröffentlichung einer Research, übermittelt durch EQS News - ein Service der EQS Group.
Für den... ► Artikel lesen | |
| AMGEN | 288,90 | +2,39 % | Mesoblast: Während Amgen und Novo Nordisk den Biotech-Hype treiben, arbeitet dieser Zelltherapie-Player am operativen Durchbruch | ||
| EPIGENOMICS | 0,870 | 0,00 % | PTA-HV: Epigenomics AG: Einladung zur ordentlichen Hauptversammlung | DJ PTA-HV: Epigenomics AG: Einladung zur ordentlichen Hauptversammlung
Hauptversammlung gemäß -- 121 Abs. 4a AktG
Epigenomics AG: Einladung zur ordentlichen Hauptversammlung
Heidelberg... ► Artikel lesen | |
| NOVAVAX | 8,620 | -1,82 % | Novavax, Inc.: Novavax Reports First Quarter 2026 Financial Results and Operational Highlights | Total revenue of $140 million in the first quarter of 2026
Pfizer agreement announced in January for non-exclusive license to utilize Matrix-M® in two infectious... ► Artikel lesen | |
| STRYKER | 251,80 | -0,16 % | Stryker launches new small bone power tool for orthopedic procedures | ||
| BIOGEN | 164,12 | +1,11 % | UCB and Biogen Announce Publication in The Lancet of Positive Dapirolizumab Pegol (DZP) Phase 3 Study Results in Systemic Lupus Erythematosus | Publication adds to previously reported findings from Phase 3 PHOENYCS GO study, demonstrating statistically significant improvement in disease activity at Week 48 with dapirolizumab pegol plus standard... ► Artikel lesen | |
| BIOFRONTERA | 2,580 | +2,38 % | PTA-News: Biofrontera AG: Biofrontera berichtet über die Ergebnisse des ersten Quartals 2026 | DJ PTA-News: Biofrontera AG: BIOFRONTERA BERICHTET ÜBER DIE ERGEBNISSE DES ERSTEN QUARTALS 2026
Unternehmensmitteilung für den Kapitalmarkt
Biofrontera AG: BIOFRONTERA BERICHTET ÜBER... ► Artikel lesen | |
| HEIDELBERG PHARMA | 2,720 | +1,12 % | EQS-HV: Heidelberg Pharma AG: Bekanntmachung der Einberufung zur Hauptversammlung am 23.06.2026 in Ladenburg mit dem Ziel der europaweiten Verbreitung gemäß §121 AktG | EQS-News: Heidelberg Pharma AG
/ Bekanntmachung der Einberufung zur Hauptversammlung
Heidelberg Pharma AG: Bekanntmachung der Einberufung zur Hauptversammlung am 23.06.2026 in... ► Artikel lesen |
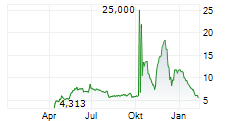
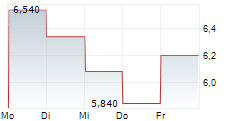
Das Unternehmen QUOIN PHARMACEUTICALS LTD ADR kann der Branche Biotechnologie zugeordnet werden. Mit einer aktuellen Kursveränderung von -0,180 (-4,81 %) liegt der Kurs bei 3,560 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu QUOIN PHARMACEUTICALS LTD ADR finden Sie auf Wallstreet Online.