- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 13:10 | Eisai whips up Alzheimer's dietary guidance to expand nutrition program beyond cancer | 3 | FiercePharma | ||
| 25.05. | Eisai legt 'konservativen' Dreijahresplan vor | 12 | Investing.com Deutsch | ||
| 25.05. | Eisai's new three-year plan seen as "conservative" | 7 | Investing.com | ||
| 25.05. | Eisai Expects Revenue Of JPY 1 Trillion For 2028 | 437 | AFX News | TOKYO (dpa-AFX) - Eisai Co., Limited (ESALY.PK, ESALF.PK, 4523.T, EII.SG), a Japanese pharmaceutical company, said on Monday that it expects revenue of JPY 1 trillion for fiscal 2028, helped... ► Artikel lesen | |
| 25.05. | BioArctic's partner Eisai presents sales simulation of potential future sales of Leqembi at its Investor Relations Day | 930 | PR Newswire | STOCKHOLM, May 25, 2026 /PRNewswire/ -- BioArctic AB's (publ) (NASDAQ Stockholm: BIOA B) partner Eisai today published a presentation including a simulation[1] of potential future sales for... ► Artikel lesen | |
| EISAI CO LTD ADR Aktie jetzt für 0€ handeln | |||||
| 21.05. | Eisai Deepens Body of Clinical Evidence for LENVIMA(R) (Lenvatinib) Across Established Indications at ASCO 2026 | 539 | JCN Newswire | TOKYO, May 21, 2026 - (JCN Newswire) - Eisai Co., Ltd. (Headquarters: Tokyo, CEO: Haruo Naito, "Eisai") announced today the presentation of clinical research across its oncology portfolio and pipeline... ► Artikel lesen | |
| 15.05. | Eisai's slow push toward blockbuster Leqembi sales gains steam with $900M forecast | 11 | FiercePharma | ||
| 15.05. | Eisai: Quartalsverlust trotz Umsatzwachstum - Gewinnprognose verfehlt | 23 | Investing.com Deutsch | ||
| 15.05. | Eisai Full-year Profit Falls, But Revenue Rises; Forecasts Higher Profit, Revenue In Fiscal 2026 | 445 | AFX News | TOKYO (dpa-AFX) - Eisai Co Ltd (ESALY.PK) on Friday reported lower profit for fiscal 2025, while revenue increased from the previous year.Profit before tax fell 16.5% to 51.0 billion yen from... ► Artikel lesen | |
| 15.05. | BioArctic: Eisai projects Leqembi revenue to total JPY 143.5 billion for fiscal year 2026 (April 2026 - March 2027) | 360 | PR Newswire | STOCKHOLM, May 15, 2026 /PRNewswire/ -- BioArctic AB's (publ) (Nasdaq Stockholm: BIOA B) partner Eisai today announced that Leqembi sales are expected to total JPY 143.5 billion (approximately... ► Artikel lesen | |
| 08.05. | Eisai, Biogen face delay to subcutaneous Leqembi | 4 | pharmaphorum | ||
| 08.05. | Eisai, Biogen Say FDA Extends Review Of LEQEMBI IQLIK As A Starting Dose For Early AD | 407 | AFX News | WESTON (dpa-AFX) - Eisai Co. Ltd (ESALY.PK) and Biogen Inc. (BIIB) on Friday said the U.S. Food and Drug Administration has extended by three months the review of LEQEMBI IQLIK as a starting... ► Artikel lesen | |
| 08.05. | FDA verlängert Prüffrist für Alzheimer-Medikament von Eisai und Biogen | 15 | Investing.com Deutsch | ||
| 08.05. | FDA extends review period for Eisai, Biogen Alzheimer's drug | 4 | Investing.com | ||
| 08.05. | Eisai Co., Ltd.: Update on FDA Priority Review of LEQEMBI IQLIK (lecanemab-irmb) Subcutaneous Injection as a Starting Dose for Early Alzheimer's Disease | 898 | PR Newswire | TOKYO and CAMBRIDGE, Mass., May 8, 2026 /PRNewswire/ -- Eisai Co., Ltd. and Biogen Inc. (Nasdaq: BIIB) today announced that the U.S. Food and Drug Administration... ► Artikel lesen | |
| 22.04. | GHIT Fund: Total Investment of Approx. USD 8.5 Million in Malaria and NTD R&D Projects With Partners Including MMV, GSK, Tanabe Pharma, Eisai and DNDi | 1.014 | PR Newswire | TOKYO, April 22, 2026 /PRNewswire/ -- The Global Health Innovative Technology (GHIT) Fund announced today a total investment of approximately JPY 1.37 billion (USD 8.5 million1) in five R&D... ► Artikel lesen | |
| 21.04. | Tortugas debuts with $106M and brain drugs from Hansoh, Eisai | 3 | BioPharma Dive | ||
| 21.04. | Merck, Eisai trial fails to improve kidney cancer treatment | 14 | Investing.com | ||
| 21.04. | MSD, Eisai report setback in first-line kidney cancer trial | 9 | pharmaphorum | ||
| 21.04. | Merck & Co., Inc., Rahway, NJ, USA and Eisai Provide Update on Phase 3 LITESPARK-012 Trial Evaluating First-Line Combination Treatments for Certain Patients With Advanced Renal Cell Carcinoma (RCC) | 579 | JCN Newswire | TOKYO and RAHWAY, N.J., Apr 21, 2026 - (JCN Newswire) - Merck & Co., Inc., Rahway, NJ, USA (known as MSD outside of the United States and Canada), and Eisai (Headquarters: Tokyo, CEO: Haruo Naito) today... ► Artikel lesen |
1 2 3 4 5 Weiter >>
119 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BAYER | 33,350 | -5,07 % | Bayer-Aktie: Das könnte brandgefährlich werden | Die Bayer-Aktie pendelte in den letzten drei Monaten in einem Seitwärtstrend zwischen 36 und 41 € auf und ab. Doch nun könnte eine Nachricht die Aktie aus diesem Trend nach unten reißen. Wie sollten... ► Artikel lesen | |
| NOVO NORDISK | 37,700 | -1,18 % | Vorsicht bei BioNTech! Novo Nordisk gibt Gas! Kaufchance bei Vidac Pharma Aktie! | Kaufstimmung bei Novo Nordisk. Die Aktie des dänischen Pharmagiganten scheint den seit Sommer 2024 begonnenen Abwärtstrend zu verlassen. Anleger reagieren auf positive Meldungen. Bei Vidac Pharma ist... ► Artikel lesen | |
| PFIZER | 22,020 | -0,07 % | Pfizer: Gedämpfte Erwartungen in der Krebsforschung lasten auf der Aktie | Pfizer bleibt unter Druck, denn Barclays sieht trotz laufender Krebsstudien kaum Chancen auf eine Kursrally. Die Analysten halten das Potenzial der Onkologie-Pipeline für begrenzt und bestätigen ihr... ► Artikel lesen | |
| NOVARTIS | 125,94 | +0,27 % | Mesoblast: Während Amgen und Novo Nordisk den Biotech-Hype treiben, arbeitet dieser Zelltherapie-Player am operativen Durchbruch | ||
| GILEAD SCIENCES | 112,40 | -0,27 % | Während Merck, Amgen, Novo Nordisk und Fresenius Milliarden bewegen, wächst die Fantasie im Zelltherapie-Sektor | ||
| AURORA CANNABIS | 3,144 | +0,58 % | War das erst der Anfang? Warum der Aurora-Deal und ein 50-Millionen-AUD-Vertrag Bioxyne weiteren Schub geben könnten | ||
| GSK | 21,130 | -0,56 % | GSK erzielt teilweisen Therapieerfolg mit Hepatitis-B-Wirkstoff | DJ GSK erzielt teilweisen Therapieerfolg mit Hepatitis-B-Wirkstoff
Von Adria Calatayud
DOW JONES--GSK hat mit einem Medikamentenkandidaten gegen Hepatitis B einen teilweisen Therapieerfolg... ► Artikel lesen | |
| ABBVIE | 183,30 | +0,11 % | AbbVie Announces European Commission Approval of AQUIPTA (atogepant) for the Acute Treatment of Migraine in Adults | AQUIPTA (atogepant) is now approved in the European Union for the acute treatment of migraine in adults, with or without aura, and for the prophylaxis of migraine in adults who have at least... ► Artikel lesen | |
| ROCHE | 347,70 | -0,18 % | Aktien Europa: Leichte Verluste - fragile Waffenruhe im Iran mahnt zur Vorsicht | PARIS/LONDON/ZÜRICH (dpa-AFX) - Die europäischen Aktienmärkte haben am Donnerstag leicht nachgegeben. Die fragile Waffenruhe im Iran-Krieg sorgte für Zurückhaltung. So hatte das US-Militär nahe der... ► Artikel lesen | |
| CANOPY GROWTH | 0,935 | +0,45 % | Canopy Growth Corporation: Canopy Growth Relaunches Tweed Brand in Germany with New MTL Cannabis Strain Lineup, Marking First International Release following Acquisition | Pablo's Revenge, Dante'z Inferno, and Frost'd Flakes launched under Tweed brand Up to five additional strains expected to follow in June 2026 Company targeting rapid growth in Germany's medical... ► Artikel lesen | |
| STADA ARZNEIMITTEL | - | - | Enzymatica AB: Quarterly report Q1/2026 - Enzymatica strengthens international position through STADA agreement | First quarter
Net sales amounted to SEK 11.4 million (12.3).
Operating result amounted to SEK -13.6.million (-18.0)
Earnings per share, before... ► Artikel lesen | |
| ELI LILLY | 929,40 | -0,10 % | HBM Healthcare Investments AG: Eli Lilly übernimmt HBM-Portfoliounternehmen Curevo für bis zu USD 1.5 Milliarden | HBM Healthcare Investments AG
/ Schlagwort(e): Ankauf
Eli Lilly übernimmt HBM-Portfoliounternehmen Curevo für bis zu USD 1.5 Milliarden
26.05.2026 / 13:10 CET/CEST
Die... ► Artikel lesen | |
| ASTRAZENECA | 155,00 | +0,32 % | Astrazeneca liefert FDA neue Daten für US-Zulassung von Camizestrant nach | DJ Astrazeneca liefert FDA neue Daten für US-Zulassung von Camizestrant nach
Von Adria Calatayud
DOW JONES--Die US-Arzneimittelbehörde FDA verschiebt ihre Entscheidung über die Zulassung eines... ► Artikel lesen | |
| TILRAY BRANDS | 4,730 | +0,11 % | Tilray Brands, Inc.: BrewDog verwandelt die diesjährige Fußball-WM in ein landesweites Sommerfest rund um Bier, Bars und große Spielmoment | BrewDog startet die Multichannel-Kampagne "Underdog" im Einzelhandel, in Bars und in digitalen Medien - einschließlich des landesweiten Gewinnspiels "Win a Year of Beer" und öffentlicher Public-Viewing-Events... ► Artikel lesen | |
| TEVA | 29,800 | 0,00 % | Teva Pharmaceutical Industries Ltd: Teva to Present at the Goldman Sachs Global Healthcare Conference | PARSIPPANY, N.J., and TEL AVIV, Israel, June 01, 2026 (GLOBE NEWSWIRE) -- Teva Pharmaceutical Industries Ltd. (NYSE and TASE: TEVA) today announced that Richard Francis, Teva's President and CEO, will... ► Artikel lesen |
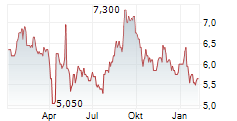
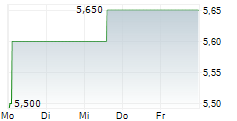
Das Unternehmen EISAI CO LTD ADR kann der Branche Pharma zugeordnet werden. Der aktuelle Kurs liegt bei 4,820 Euro und damit -0,41 % im Minus.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu EISAI CO LTD ADR finden Sie auf Wallstreet Online.