- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| Mo | Nxera Pharma Announces Progress of Out-Licensing of GPCR-targeted Program and Participation in Series A Financing | 229 | GlobeNewswire (Europe) | New company ("NewCo") co-founded by Nxera with a syndicate of leading healthcare and life sciences-focused investment firms.In return for granting an exclusive worldwide license to the program, Nxera... ► Artikel lesen | |
| 08.05. | Nxera Pharma verklagt OMass Therapeutics wegen Patentverletzung | 4 | Investing.com Deutsch | ||
| 08.05. | Nxera Pharma sues OMass Therapeutics over patent infringement | 2 | Investing.com | ||
| 08.05. | Nxera Pharma: Nxera Files Patent Infringement Action in Relation to its miniG Platform Technology | 1.115 | GlobeNewswire (Europe) | Tokyo, Japan and Cambridge and London, UK, 8 May 2026 - Nxera Pharma Co., Ltd. ("Nxera Pharma" or "the Company"; TSE 4565) announces that its wholly owned subsidiary Nxera Pharma UK Limited ("Nxera")... ► Artikel lesen | |
| 01.05. | Nxera Pharma Operational Highlights and Consolidated Results for the First Quarter 2026 | 193 | GlobeNewswire (Europe) | Tokyo, Japan and Cambridge, UK, 1 May 2026 - Nxera Pharma ("the Company" or "Nxera"; TSE: 4565) provides an update on operational activities and reports its consolidated results for the first quarter... ► Artikel lesen | |
| 20.04. | Nxera Pharma To Receive $10 Mln Milestone Payment From AbbVie Under Neurology Collaboration | 366 | AFX News | TOKYO (dpa-AFX) - Nxera Pharma Co., Ltd. (4565.T, SOLTF), a research-based biopharmaceutical company, on Monday announced that it will receive a $10 million milestone payment from AbbVie Inc.... ► Artikel lesen | |
| 20.04. | Nxera Pharma to Receive US$10 Million Milestone Payment from AbbVie under Collaboration Targeting Neurological Diseases | 168 | GlobeNewswire (Europe) | This marks the third milestone achieved under the collaboration and reflects the steady progress of the pipelineUnder the terms of the agreement, Nxera is eligible to receive up to US$1.2 billion... ► Artikel lesen | |
| NXERA PHARMA Aktie jetzt für 0€ handeln | |||||
| 16.04. | TM Nxera Tops Out AI-Ready Johor Data Centre, Moves Towards 2H2026 Ops | 1 | BusinessToday | ||
| 15.04. | Nxera Pharma: Notice of Issuance of New Shares in Connection with the Introduction of the Employee Stock Ownership Plan, Matters Relating to Stock Units and Determination of Payment Amount and Other Matters of Issuance of New Shares Under Prior ... | 3 | GlobeNewswire (USA) | ||
| 14.04. | Nxera Pharma's QUVIVIQ (daridorexant) 25mg and 50mg Receives Approval in Taiwan for the Treatment of Insomnia | 422 | GlobeNewswire (Europe) | Nxera's partner Holling Bio-Pharma Corp. will commercialize QUVIVIQ® in Taiwan, a market with approximately 4-5 million patients, with launch planned during 2026QUVIVIQ® is approved through Taiwan's... ► Artikel lesen | |
| 14.04. | TM Nxera tops out its first AI data centre in Johor | - | Developing Telecoms | ||
| 13.04. | Singtel's Nxera and Telekom Malaysia top out data center in Johor | 2 | DatacenterDynamics | ||
| 13.04. | Nxera Pharma's Partner Neurocrine Biosciences Doses First Patient in Phase 2 Trial of NBI-1117570 for Adults with Schizophrenia | 282 | GlobeNewswire (Europe) | NBI-1117570 is an investigational oral, dual muscarinic M1/M4 selective receptor agonist discovered by Nxera advancing through clinical development under a multi-program collaboration with NeurocrineNxera... ► Artikel lesen | |
| 09.04. | Nxera Pharma Achieves Second Development Milestone in Collaboration with Eli Lilly Targeting Metabolic Diseases | 552 | GlobeNewswire (Europe) | Tokyo, Japan and Cambridge, UK, 09 April 2026 - Nxera Pharma Co., Ltd. ("Nxera" or "the Company"; TSE 4565) today announces that it has achieved a second development milestone under its multi-target... ► Artikel lesen | |
| 01.04. | Nxera Pharma's Partner Centessa Pharmaceuticals to be Acquired by Lilly | 232 | GlobeNewswire (Europe) | Lilly to gain ownership of Centessa's OX2R agonist pipeline designed to improve outcomes across sleep-wake disordersCentessa's OX2R agonist pipeline was jointly discovered by Nxera and Centessa, and... ► Artikel lesen | |
| 05.03. | Nxera Pharma to Receive US$3 Million Milestone Payment from Centessa Pharmaceuticals | 265 | GlobeNewswire (Europe) | Tokyo, Japan and Cambridge, UK, 5 March 2026 - Nxera Pharma Co., Ltd. ("Nxera" or "the Company"; TSE 4565) today announces that Centessa Pharmaceuticals has achieved an additional early development... ► Artikel lesen | |
| 04.03. | Nxera Pharma Co Files South Korea Marketing Application For Daridorexant | - | RTTNews | ||
| 04.03. | Nxera Pharma Submits Marketing Authorization Application for Daridorexant in South Korea | 1 | GlobeNewswire (USA) | ||
| 13.02. | Nxera Pharma reports Q4 results | 1 | Seeking Alpha | ||
| 13.02. | Nxera Pharma Proposes Changes to its Board of Directors | 2 | GlobeNewswire (USA) |
1 2 3 Weiter >>
60 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BAYER | 37,710 | -1,23 % | Tagesvorschau 12. Mai: Quartalsflut am Dienstag: Bayer, Munich Re und Siemens Energy öffnen ihre Bücher | Am Dienstag prasseln Quartalszahlen, Hauptversammlungen und brisante Konjunkturdaten gleichzeitig auf die Märkte ein - Anleger sollten wach bleiben.Dieser Dienstag verlangt Anlegern volle Aufmerksamkeit:... ► Artikel lesen | |
| PFIZER | 21,780 | -0,07 % | Pfizer-Aktie: Bleibt für Dividenden-Anleger attraktiv | Die Aktie von Pfizer hat in den vergangenen Monaten deutlich besser abgeschnitten als viele andere Werte aus dem Gesundheitssektor. Trotz anhaltender Skepsis gegenüber Pharmaunternehmen mehren sich... ► Artikel lesen | |
| NOVARTIS | 127,34 | +0,08 % | Biotech und die Effizienz-Revolution: Warum für Novartis, BioNTech und BioNxt die letzte Meile entscheidet | Während für Biotech-Investoren über Jahrzehnte hinweg das Dogma galt, dass nur die Entdeckung eines völlig neuen Moleküls den Weg zu einem Milliarden-Exit ebnen kann, zeigt die Realität des Marktes... ► Artikel lesen | |
| GILEAD SCIENCES | 111,50 | -0,05 % | Gilead Sciences, Inc.: Gilead to Present New Data Advancing Care in PBC and Viral Hepatitis at EASL 2026 | - Late-Breaking Livdelzi (Seladelpar) Data Expand the Evidence Base in PBC, while Interim Data Show Durable, Consistent Results Across Key Markers - - Bulevirtide Data Strengthen Evidence of... ► Artikel lesen | |
| SANOFI | 73,83 | +0,12 % | Mesoblast Aktie: Zelltherapie im Aufwind - warum der kleine Biotech-Player plötzlich in einer Liga mit Merck, Regeneron & Co. gedacht werden muss | ||
| ABBVIE | 181,50 | +0,22 % | Which Pharma Giant Offers Better Growth: AstraZeneca or AbbVie? | ||
| ROCHE | 352,00 | +0,91 % | Roche-Chef macht Anlegern Mut: Roche kündigt die nächste Wachstumswelle an - drei Medikamente im Fokus | © Foto: Georgios Kefalas/KEYSTONE/dpaRoche verspricht Wachstum weit über 2030 hinaus. Neue Medikamente und eine starke Pipeline sollen den Pharmakonzern absichern.Roche erwartet, dass die bereits auf... ► Artikel lesen | |
| ABBOTT LABORATORIES | 72,86 | +0,22 % | How Abbott's Volume-Driven Nutrition Strategy Is Shaping Up | ||
| DERMAPHARM | 51,80 | +2,57 % | Ihre wichtigsten Termine: Frische Q-Zahlen von Cisco, Porsche, Dermapharm, Siemens, Deutsche Telekom & Eon | © Foto: Thiago Prudencio/DAX via ZUMA Press Wire/dpaGut geplant in den Tag. Mit dem wO-Tagesausblick haben Sie die wichtigsten Termine im Blick und starten bestens vorbereitet in den neuen Handelstag.Unternehmenstermine... ► Artikel lesen | |
| ZOETIS | 63,82 | -0,13 % | ZOETIS INC: Stabile Phase mit klarer Struktur | ||
| DOCMORRIS | 7,675 | -1,85 % | Ihre wichtigsten Termine: Alle Blicke auf: 3M, Bayer, Thyssenkrupp, DocMorris, Rheinmetall und Lufthansa! | © Foto: Daniel Bockwoldt/dpaGut geplant in den Tag. Mit dem wO-Tagesausblick haben Sie die wichtigsten Termine im Blick und starten bestens vorbereitet in den neuen Handelstag.Unternehmenstermine 06:55... ► Artikel lesen | |
| HAEMATO | 11,700 | -4,10 % | M1 Kliniken schließt Verkauf von Haemato Pharm ab | Die M1 Kliniken AG hat den Verkauf der Haemato Pharm GmbH über ihre Mehrheitsbeteiligung Haemato AG an die Phoenix Group mit Wirkung zum Ablauf des 31. Januar 2026 abgeschlossen. Sämtliche Vollzugsbedingungen... ► Artikel lesen | |
| INCYTE | 82,74 | +0,88 % | Incyte gibt bekannt, dass mehr als 20 Abstrakte für die Präsentation auf dem Kongress der European Hematology Association (EHA) 2026 angenommen wurden | Daten aus der zulassungsrelevanten Phase-3-Studie "frontMIND" zur Bewertung von Tafasitamab (Monjuvi/Minjuvi) bei der Erstlinienbehandlung des diffusen großzelligen B-Zell-Lymphoms (DLBCL) wurden... ► Artikel lesen | |
| SCHOTT PHARMA | 16,380 | +0,99 % | EQS-News: SCHOTT Pharma AG & Co. KGaA: SCHOTT Pharma mit robustem erstem Halbjahr und starkem Cashflow | EQS-News: SCHOTT Pharma AG & Co. KGaA
/ Schlagwort(e): Halbjahresbericht/Halbjahresergebnis
SCHOTT Pharma mit robustem erstem Halbjahr und starkem Cashflow
13.05.2026 /... ► Artikel lesen | |
| EDWARDS LIFESCIENCES | 69,96 | -0,09 % | Edwards Lifesciences Appoints Theodora Mistras As Chief Financial Officer | WASHINGTON (dpa-AFX) - Edwards Lifesciences Corporation (EW) said that it has appointed Theodora Mistras as chief financial officer effective at the end of May.Mistras will join Edwards from... ► Artikel lesen |
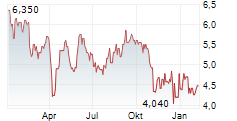
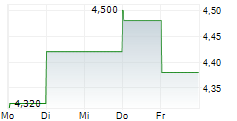
Das Unternehmen NXERA PHARMA CO LTD kann der Branche Pharma zugeordnet werden. Mit einer aktuellen Kursveränderung von -0,200 (-3,17 %) liegt der Kurs bei 6,100 Euro. Aus der Ausschüttung der letzten 12 Monate von insgesamt 0,00 Euro errechnet sich eine Dividendendrendite von 0,00 %.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu NXERA PHARMA CO LTD finden Sie auf Wallstreet Online.