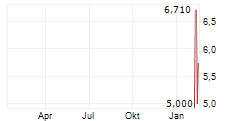| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
| 5,700 | 5,900 | 21.05. | |
| 5,830 | 5,900 | 21.05. |
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| Do | Median Technologies Announces the Availability of the Preparatory Documents for the Annual Shareholders' Ordinary and Extraordinary General Meeting on June 10, 2026 | 336 | Business Wire | Regulatory News:
The shareholders of Median Technologies (FR0011049824, ALMDT, "Median" or "The Company") are invited to participate in the Annual Shareholders' Ordinary and Extraordinary General... ► Artikel lesen | |
| MEDIAN TECHNOLOGIES Aktie jetzt für 0€ handeln | |||||
| 04.05. | Median Technologies: Disclosure of Total Number of Voting Rights and Number of Shares in the Capital as of April 30, 2026 | 249 | Business Wire | Pursuant to article L-223-8 II of the French "Code de commerce" and 223-16 of the AMF's General Regulations.
Regulatory News:
Median Technologies (Paris:ALMDT):
Total number of shares... ► Artikel lesen | |
| 23.04. | Median Technologies Reports Full-Year 2025 Financial Results and Provides Key Q1 2026 Business Highlights | 692 | Business Wire | eyonis LCS 510(k) clearance sets stage for commercialization in the United States
New leadership of U.S. subsidiary and collaboration with Tempus accelerate rollout
28% reduction... ► Artikel lesen | |
| 07.04. | Median Technologies: Disclosure of Total Number of Voting Rights and Number of Shares in the Capital as of March 31, 2026 | 346 | Business Wire | Pursuant to article L-223-8 II of the French "code de Commerce" and 223-16 of the AMF's General Regulations.
Regulatory News:
Median Technologies (Paris:ALMDT):
Total number of shares... ► Artikel lesen | |
| 18.03. | Median Technologies Achieves ISO 13485:2016 Certification for eyonis Medical Device Quality Management System | 480 | Business Wire | Certification of a robust and compliant Quality Management System for AI-based Software as Medical Devices, as per internationally recognized medical device regulatory standards
Pledge... ► Artikel lesen | |
| 13.03. | Median Technologies Meets the Eligibility Conditions for the PEA-PME Scheme | 424 | Business Wire | Regulatory News:
Median Technologies (FR0011049824, ALMDT, PEA-PME scheme eligible, "Median" or the "Company"), developer of eyonis, a suite of artificial intelligence (AI) powered Software as... ► Artikel lesen | |
| 09.03. | Median Technologies: Disclosure of Total Number of Voting Rights and Number of Shares in the Capital as of February 28, 2026 | 331 | Business Wire | Pursuant to article L-223-8 II of the French "Code de commerce" and 223-16 of the AMF's General Regulations.
Regulatory News:
Median Technologies (Paris:ALMDT):
Total number of shares... ► Artikel lesen | |
| 02.03. | Median Technologies to Showcase eyonis LCS, Its AI/ML-based Software as a Medical Device for Lung Cancer Screening, at the European Congress of Radiology | 425 | Business Wire | eyonis LCS commercial launch is ongoing in U.S. following FDA 510(k) clearance
CE-marking regulatory process is on-going with a decision expected in Q2 2026 that could trigger commercial... ► Artikel lesen | |
| 27.02. | Median Technologies to present at the TD Cowen 46th Annual Health Care Conference | 402 | Business Wire | Regulatory News:
Median Technologies (FR0011049824, ALMDT, "Median" or the "Company"), developer of eyonis, a suite of artificial intelligence (AI) powered Software as a Medical Device (SaMD) for... ► Artikel lesen | |
| 23.02. | Median Technologies to Host Webcasts Highlighting Strategic Advances Driving U.S. Deployment of eyonis LCS | 324 | Business Wire | Webcasts on February 26, 2026, at 5:45 pm CET (held in French): sign-up link and 6:45 pm CET/12:45 pm ET (held in English): sign-up link
Regulatory News:
Median Technologies (FR0011049824, ALMDT... ► Artikel lesen | |
| 18.02. | Median Technologies: FDA Clearance Sets Stage for U.S. Commercialization of eyonis LCS Software as a Medical Device for Lung Cancer Screening; Oran Muduroglu Appointed President of Median eyonis Inc. | 605 | Business Wire | Oran Muduroglu, proven medical imaging entrepreneur, to lead U.S. launch of eyonis LCS
Established U.S. commercial and clinical infrastructure combined with Medicare reimbursement pathway... ► Artikel lesen | |
| 12.02. | Median Technologies Announces Collaboration With Tempus to Expand Access to eyonis LCS Software as a Medical Device in the United States | 473 | Business Wire | The collaboration follows FDA 510(k) clearance for eyonis LCS and aims to integrate high-performance lung cancer screening detection and diagnosis device into the clinical workflow through the Tempus... ► Artikel lesen | |
| 11.02. | Median Technologies: Disclosure of Total Number of Voting Rights and Number of Shares in the Capital as of January 31, 2026 | 388 | Business Wire | Pursuant to article L-223-8 II of the French "code de Commerce" and 223-16 of the AMF's General Regulations.
Regulatory News:
Median Technologies (Paris:ALMDT):
Total number of shares... ► Artikel lesen | |
| 09.02. | Median Technologies receives FDA clearance for lung cancer AI tool | 8 | Investing.com | ||
| 09.02. | Median Technologies Receives FDA 510(k) Clearance for eyonis LCS, the First AI Tech-Based Detection and Diagnosis Device for Lung Cancer Screening | 1.335 | Business Wire | eyonis LCS aims to redefine lung cancer screening by supporting diagnosis at early, curable stages, while reducing false positives to avoid unnecessary follow-up procedures, and has the potential... ► Artikel lesen | |
| 19.01. | Median Technologies Reports Landmark 2025 Performance With Major Regulatory and Commercial Milestones Ahead in 2026 | 424 | Business Wire | Record iCRO bookings in 2025; order backlog at an all-time high of €76.6 million despite negative forex impact of (6.5) million
2025 revenue of €23.5 million, up 2.6% vs 2024 despite negative... ► Artikel lesen | |
| 14.01. | Median Technologies: H1 2026 Financial Communication Calendar | 407 | Business Wire | Regulatory News:
Median Technologies (FR0011049824, ALMDT, PEA-PME scheme eligible, "Median" or the "Company"), manufacturer of eyonis, a suite of artificial intelligence (AI) powered Software... ► Artikel lesen | |
| 07.01. | Half Year Median Technologies Liquidity Contract Statement | 669 | Business Wire | Regulatory News:
Under the liquidity contract entrusted by Median Technologies (FR0011049824, ALMDT, PEA/SME eligible, "Median" or "the Company") to TP ICAP (Europe), the following resources were... ► Artikel lesen | |
| 05.01. | Median Technologies: Disclosure of Total Number of Voting Rights and Number of Shares in the Capital as of December 31, 2025 | 341 | Business Wire | Pursuant to article L-223-8 II of the French "code de Commerce" and 223-16 of the AMF's General Regulations.
Regulatory News:
Median Technologies (Paris:ALMDT):
Total number of shares... ► Artikel lesen | |
| 08.12.25 | Median Technologies Partners With Leading U.S. Healthcare Company to Bring eyonis LCS to Patients Across the U.S. and Europe | 402 | Business Wire | Non-exclusive commercial agreement for the distribution of eyonis LCS in the United States and Europe
The agreement will become effective upon FDA 510(k) clearance
Partner's robust... ► Artikel lesen |
1 2 3 Weiter >>
41 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| PALANTIR TECHNOLOGIES | 118,36 | +0,36 % | Aktienanalysen der Woche: D-Wave Quantum, Palantir & Cisco Systems | ||
| ORACLE | 163,36 | +2,91 % | Oracle: Wie stabil ist diese Erholung wirklich? | Die Oracle-Aktie hat sich nach einer schwächeren Phase zuletzt spürbar stabilisiert. Im Fokus steht nun, ob die aktuelle technische Struktur Bestand haben kann Den vollständigen Artikel lesen ... ► Artikel lesen | |
| SHOPIFY | 90,57 | +0,27 % | PayPal: Shopify greift frontal an | Für PayPal kommt der nächste Gegenwind aus einer Richtung, die Anleger unbedingt im Blick haben sollten. Der E-Commerce-Spezialist Shopify baut sein Payment-Geschäft in den USA weiter aus und hat dafür... ► Artikel lesen | |
| CROWDSTRIKE | 557,20 | -0,38 % | 400 Prozent in 9 Tagen mit CrowdStrike: Heute neue Chance | Im HEBELTRADER geht es derzeit Schlag auf Schlag. Erst vergangene Woche wurde bei CrowdStrike nach nur 9 Kalendertagen bereits das erste Kursziel erreicht - mit einem Gewinn von 400 Prozent. Genau jetzt... ► Artikel lesen | |
| D-WAVE QUANTUM | 22,190 | +33,84 % | D-Wave Quantum-Aktie +25%: Startet jetzt die nächste Quanten-Rallye? | Nach einer kräftigen Erholung ist die Aktie von D-Wave Quantum im Anschluss an die jüngsten Quartalszahlen wieder deutlich zurückgefallen. Doch am Donnerstag schießt der Kurs plötzlich um gut +25% in... ► Artikel lesen | |
| ATOSS SOFTWARE | 78,90 | -1,74 % | DAX-Check LIVE: ATOSS Software, Hochtief, Ottobock, Renk, SAP, SFC Energy im Fokus | Der deutsche Aktienmarkt hat am Dienstag mit moderaten Gewinnen geschlossen. Der DAX knüpfte zunächst an die kräftige Erholungsbewegung vom Wochenauftakt an und legte im Verlauf bis zu 1,5 Prozent zu.... ► Artikel lesen | |
| ZOOM COMMUNICATIONS | 83,50 | -2,32 % | Ihre wichtigsten Termine: Spannende Zahlen von: Zoom Video Communications, Walmart, Deere & Co und Easyjet | © Foto: Nam Y. Huh/AP/dpaGut geplant in den Tag. Mit dem wO-Tagesausblick haben Sie die wichtigsten Termine im Blick und starten bestens vorbereitet in den neuen Handelstag.Unternehmenstermine 07:00... ► Artikel lesen | |
| VEEVA SYSTEMS | 136,15 | -4,02 % | Veeva Systems (VEEV) Rises Yet Lags Behind Market: Some Facts Worth Knowing | ||
| BRAINCHIP | 0,092 | -1,92 % | BrainChip Holdings Ltd: BrainChip Enables the Next Generation of Always-On Wearables With the AkidaTag© Reference Platform | New Platform for wearable AI and industrial uses combines the company's Akida AKD1500 neuromorphic co-processor silicon with Nordic Semiconductor's nRF5340 wireless SoC for intelligent low-power... ► Artikel lesen | |
| GEN DIGITAL | 21,450 | -0,23 % | Dividendenbekanntmachungen (18.05.2026) | Unternehmen ISIN-Code Dividende (Währung) Dividende (EUR) A2A SPA IT0001233417 - 0,104 EUR AIR LIQUIDE SA FR0000120073 - 3,7 EUR ALCOA CORPORATION CDIS AU0000339426 0,1 USD 0,086 EUR ALLISON... ► Artikel lesen | |
| VERSES AI | 0,540 | +6,93 % | VERSES AI Inc.: VERSES AI Corporation Restructures to Focus on Core Target Market | VANCOUVER, British Columbia, Jan. 16, 2026 (GLOBE NEWSWIRE) -- VERSES AI Inc. (CBOE: VERS) (OTCQB: VRSSF) ("VERSES' or the "Company"), a cognitive computing company specializing in next-generation... ► Artikel lesen | |
| GRAB | 3,073 | +1,96 % | Grab Holdings To Consolidate Superbank After Stake Transfer To GXS Bank | SINGAPUR (dpa-AFX) - Grab Holdings Ltd. (GRAB), on Wednesday announced plans to consolidate PT Super Bank Indonesia Tbk.Singtel Alpha Investments Pte. Ltd. will transfer its stake in Superbank... ► Artikel lesen | |
| SENTINELONE | 15,575 | +0,81 % | SentinelOne (S) Laps the Stock Market: Here's Why | ||
| TREND MICRO | 32,680 | -1,36 % | Trend Micro Incorporated: Trend Micro Reports Earnings Results for Q1 2026 | Increase of 9% YoY in total company net sales*, driving U.S. $1.7 Billion in ARR** with operating margin exceeding plan at 21%
Increase of 50% YoY in TrendAI... ► Artikel lesen | |
| MITEK SYSTEMS | 13,500 | +5,97 % | Mitek and Synectics Solutions Partner to Help UK Insurers Tackle Rising Digital Fraud While Expanding Fair Access to Insurance | Real-time identity verification and consortium fraud intelligence combined to improve onboarding risk decisions
Mitek (NASDAQ: MITK) and Synectics Solutions have partnered to help UK insurers detect... ► Artikel lesen |
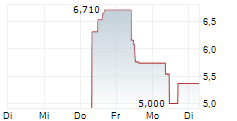
Das Unternehmen MEDIAN TECHNOLOGIES kann der Branche Software zugeordnet werden. Die relative Kursveränderung beträgt aktuell +6,35 % bei einem Aktienkurs von 5,860 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu MEDIAN TECHNOLOGIES finden Sie auf Wallstreet Online.