| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
| 0,079 | 0,086 | 05.06. |
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 20.05. | GenSight Biologics Announces the Results of its Combined General Meeting of May 19, 2026 | 335 | Business Wire | Regulatory News:
GenSight Biologics (Euronext: SIGHT, ISIN: FR0013183985, PEA-PME eligible), a biopharma company focused on developing and commercializing innovative gene therapies for retinal... ► Artikel lesen | |
| 18.05. | GenSight Biologics S.A.: GenSight Biologics to Hold its Combined General Meeting on May 19, 2026 | 366 | Business Wire | Combined General Meeting on May 19, 2026
Key operational developments in 2025 and 2026
Regulatory News:
GenSight Biologics (the "Company") (Euronext: SIGHT, ISIN: FR0013183985, PEA-PME... ► Artikel lesen | |
| 11.05. | GenSight Biologics S.A.: GenSight Biologics Announces Publication on Matching-Adjusted Indirect Comparison of Leber Hereditary Optic Neuropathy Treatments | 294 | Business Wire | Regulatory News:
GenSight Biologics (Euronext: SIGHT, ISIN: FR0013183985, PEA-PME eligible), a biopharma company focused on developing and commercializing innovative gene therapies for retinal... ► Artikel lesen | |
| 28.04. | GenSight Biologics S.A.: GenSight Biologics Announces Annual General Meeting to Take Place on May 19, 2026 | 556 | Business Wire | Procedures for obtaining preparatory documents for the General Meeting are described below.
Regulatory News:
GenSight Biologics (Euronext: SIGHT, ISIN: FR0013183985, PEA-PME eligible), a biopharma... ► Artikel lesen | |
| GENSIGHT BIOLOGICS Aktie jetzt für 0€ handeln | |||||
| 15.04. | GenSight Biologics Announces the Filing of its 2025 Universal Registration Document | 348 | Business Wire | Regulatory News:
GenSight Biologics (Euronext: SIGHT, ISIN: FR0013183985, PEA-PME eligible), a biopharma company focused on developing and commercializing innovative gene therapies for retinal... ► Artikel lesen | |
| 07.04. | GenSight Biologics Reports Cash Position and Revenue from Early Access Program as of March 31, 2026 | 417 | Business Wire | Regulatory News:
GenSight Biologics (Euronext: SIGHT, ISIN: FR0013183985, PEA-PME eligible), a biopharma company focused on developing and commercializing innovative gene therapies for retinal... ► Artikel lesen | |
| 03.04. | GenSight Biologics Confirms Definitive Full-Year 2025 Consolidated Financial Results Are in Line with Estimates | 608 | Business Wire | Regulatory News:
GenSight Biologics (Euronext: SIGHT, ISIN: FR0013183985, PEA-PME eligible), a biopharma company focused on developing and commercializing innovative gene therapies for retinal... ► Artikel lesen | |
| 27.03. | GenSight Biologics Reports Estimated Full-Year Consolidated Financial Results for 2025 | 376 | Business Wire | 2025 was a turning point with key regulatory milestones providing a solid foundation for 2026.
Cash runway is projected to extend through December 2026.
Preparation of the Phase... ► Artikel lesen | |
| 20.03. | The 15-20 National Hospital and GenSight Biologics Announce the First Treatments in the French Named Patient Early Access Program (AAC) for GS010/LUMEVOQ | 364 | Business Wire | The first group of patients in the GS010 AAC program were treated at the 15-20 National Hospital in Paris on March 19.
The REVISE dose-ranging study is proceeding according to the planned... ► Artikel lesen | |
| 18.03. | GenSight Biologics Announces the Closing of its Nearly €1.7 Million Fundraising | 457 | Business Wire | Regulatory News:
NOT FOR DISTRIBUTION, DIRECTLY OR INDIRECTLY, IN THE UNITED STATES OF AMERICA, CANADA, AUSTRALIA, JAPAN AND SOUTH AFRICA
GenSight Biologics (the "Company") (Euronext: SIGHT... ► Artikel lesen | |
| 10.03. | GenSight Biologics: €1.7 Million Raised Through Share And Warrant Financing To Extend Cash Runway | 1 | pulse2.com | ||
| 09.03. | GenSight Biologics Provides Updates about GS010/LUMEVOQ Early Access Programs and the Ongoing REVISE Study | 378 | Business Wire | Regulatory News:
GenSight Biologics (Euronext: SIGHT, ISIN: FR0013183985, PEA-PME eligible), a biopharma company focused on developing and commercializing innovative gene therapies for retinal... ► Artikel lesen | |
| 18.02. | GenSight Biologics S.A.: GenSight Biologics Bolsters Regulatory Leadership in US and Europe with Two Senior Appointments | 419 | Business Wire | Regulatory News:
GenSight Biologics ("GenSight Biologics" or the "Company") (Euronext: SIGHT, ISIN: FR0013183985, PEA-PME eligible), a biopharma company focused on developing and commercializing... ► Artikel lesen | |
| 10.02. | GenSight Biologics S.A.: The 15-20 National Hospital and GenSight Biologics Announce the Treatment of the First Patient in the GS010/LUMEVOQ REVISE Study | 483 | Business Wire | First of 14 planned patients for the dose-ranging study approved by the ANSM in December 2025
Clinical study marks continuing partnership between the 15-20 Hospital and GenSight Biologics... ► Artikel lesen | |
| 29.01. | GenSight Biologics S.A.: GenSight Biologics Announces the Results of its Extraordinary General Meeting of January 28, 2026 | 950 | Business Wire | Regulatory News:
GenSight Biologics (Euronext: SIGHT, ISIN: FR0013183985, PEA-PME eligible), a biopharma company focused on developing and commercializing innovative gene therapies for retinal... ► Artikel lesen | |
| 08.01. | GenSight Biologics Reports Cash Position as of December 31, 2025, and Provides Business Updates | 494 | Business Wire | Cash position amounted to €2.4 million as of December 31, 2025
Successful closing of fundraising worth nearly €2.9 million on January 7, 2026
Payments for early access treatments... ► Artikel lesen | |
| 07.01. | GenSight Biologics Announces Closing of Fundraising Worth Nearly EUR 2.9 Million | 457 | Business Wire | Regulatory News:
NOT FOR DISTRIBUTION, DIRECTLY OR INDIRECTLY, IN THE UNITED STATES OF AMERICA, CANADA, AUSTRALIA, JAPAN AND SOUTH AFRICA
GenSight Biologics (the "Company") (Euronext: SIGHT... ► Artikel lesen | |
| 29.12.25 | GenSight Biologics Announces Successful Fundraising Amounting to Nearly €2.9 Million | 492 | Business Wire | Regulatory News:
NOT FOR DISTRIBUTION, DIRECTLY OR INDIRECTLY, IN THE UNITED STATES OF AMERICA, CANADA, AUSTRALIA, JAPAN AND SOUTH AFRICA
GenSight Biologics ("GenSight" or the "Company") (Euronext:... ► Artikel lesen | |
| 22.12.25 | GenSight Biologics S.A.: GenSight Biologics Announces Regulatory Authorization for Early Access Treatment with GS010/LUMEVOQ in Israel | 567 | Business Wire | Israel's Ministry of Health authorizes individual patients early access treatment with GS010/LUMEVOQ, a candidate gene therapy for the treatment of ND4-LHON.
Bilateral injection expected... ► Artikel lesen | |
| 22.12.25 | GenSight Biologics S.A.: GenSight Biologics Announces the Granting of Compassionate Use Authorization (CUA/AAC) for GS010/LUMEVOQ in France | 595 | Business Wire | French medicines agency ANSM has granted authorization for named patient early access program for GS010/LUMEVOQ, a gene therapy indicated for the treatment of ND4-LHON.
Regulatory News:
GenSight... ► Artikel lesen |
1 2 Weiter >>
37 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BIONTECH | 77,05 | -1,34 % | BioNTech SE: Globale Daten der ROSETTA Lung-02-Studie: Ermutigende Wirksamkeit für BioNTechs und Bristol Myers Squibbs bispezifischen PD-L1xVEGF-A-Immunmodulator-Kandidat Pumitamig bei nicht-kleinzelligem Lungenkrebs | Erster bispezifischer PD-(L)1xVEGF-Immunmodulator-Kandidat, zu dem globale Daten mit ermutigender Wirksamkeit in Kombination mit Chemotherapie in der Erstlinienbehandlung von nicht-kleinzelligem Lungenkrebs... ► Artikel lesen | |
| EVOTEC | 4,932 | -3,58 % | EQS-News: Evotec SE: Evotec ernennt Rui Wang zum Head of Global In Silico & AI | EQS-News: Evotec SE
/ Schlagwort(e): Sonstiges
Evotec ernennt Rui Wang zum Head of Global In Silico & AI
02.06.2026 / 07:30 CET/CEST
Für den Inhalt der Mitteilung ist... ► Artikel lesen | |
| BB BIOTECH | 45,850 | -0,86 % | BB Biotech: "Wachstumsstarke Mid- und Small Caps bilden das Rückgrat unseres Portfolios": Mehr Einzelwerte und ... | ||
| MEDIGENE | 0,025 | +5,08 % | PTA-Adhoc: Medigene AG: Widerruf der Zulassung der Aktien der Medigene AG zum Handel im regulierten Markt | DJ PTA-Adhoc: Medigene AG: Widerruf der Zulassung der Aktien der Medigene AG zum Handel im regulierten Markt
Veröffentlichung von Insiderinformationen gemäß Artikel 17 MAR Schlagwort(e): Sonstiges... ► Artikel lesen | |
| QIAGEN | 31,920 | -2,18 % | DEUTSCHE BANK RESEARCH stuft QIAGEN NV auf 'Buy' | FRANKFURT (dpa-AFX Analyser) - Deutsche Bank Research hat Qiagen mit einem Kursziel von 43 US-Dollar auf "Buy" belassen. Das Feedback aus Gesprächen mit etlichen US-Laborexperten deute darauf hin, dass... ► Artikel lesen | |
| MODERNA | 41,190 | +0,12 % | Opening Bell: Bitcoin, Strategy, Broadcom, Marvell, Micron, Axon Enterprise, Moderna, Lululemon | Die US-Börsen haben am Donnerstag eine zweigeteilte Entwicklung gezeigt. Der Dow Jones eröffnete mit klaren Gewinnen, baute diese im Verlauf deutlich aus und schloss 1,73 Prozent fester. Der Nasdaq... ► Artikel lesen | |
| PAION | 0,062 | -23,60 % | XFRA MISTRADE ANTRAG IN ISIN DE000A3E5EG5 WURDE STATTGEGEBEN | Dem Mistrade-Antrag wurde stattgegeben. Folgende Geschaefte in der ISIN DE000A3E5EG5, Wertpapier-Name: PAION AG INH O.N., wurden aufgehoben:ISIN Datum Zeit Volumen PreisDE000A3E5EG5 02.02.2026 08:13:40 300 0... ► Artikel lesen | |
| VALNEVA | 2,331 | -0,64 % | SCHOCKMELDUNG bei Valneva: Was die wenigsten Anleger auf dem Schirm haben - Was ich konkret unternommen habe | ||
| AMGEN | 306,30 | +0,97 % | Während Merck, Amgen, Novo Nordisk und Fresenius Milliarden bewegen, wächst die Fantasie im Zelltherapie-Sektor | ||
| EPIGENOMICS | 0,870 | 0,00 % | PTA-HV: Epigenomics AG: Einladung zur ordentlichen Hauptversammlung | DJ PTA-HV: Epigenomics AG: Einladung zur ordentlichen Hauptversammlung
Hauptversammlung gemäß -- 121 Abs. 4a AktG
Epigenomics AG: Einladung zur ordentlichen Hauptversammlung
Heidelberg... ► Artikel lesen | |
| NOVAVAX | 8,080 | -1,70 % | NOVAVAX INC: Stabilität vor der nächsten Bewegung | ||
| STRYKER | 265,60 | +0,15 % | Leerink cuts Stryker stock price target to $407 on model update | ||
| BIOGEN | 171,56 | +1,19 % | Biogen's Salanersen Wins FDA Breakthrough Therapy Designation For Spinal Muscular Atrophy Treatment | WESTON (dpa-AFX) - Thursday, Biogen Inc. (BIIB) announced that the U.S. Food and Drug Administration has granted salanersen Breakthrough Therapy Designation for the treatment of spinal muscular... ► Artikel lesen | |
| BIOFRONTERA | 2,490 | -3,49 % | PTA-News: Biofrontera AG: Biofrontera berichtet über die Ergebnisse des ersten Quartals 2026 | DJ PTA-News: Biofrontera AG: BIOFRONTERA BERICHTET ÜBER DIE ERGEBNISSE DES ERSTEN QUARTALS 2026
Unternehmensmitteilung für den Kapitalmarkt
Biofrontera AG: BIOFRONTERA BERICHTET ÜBER... ► Artikel lesen | |
| HEIDELBERG PHARMA | 2,710 | +1,50 % | HEIDELBERG PHARMA AG: Ruhe vor der nächsten Phase |
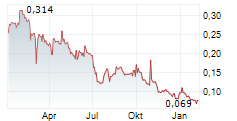
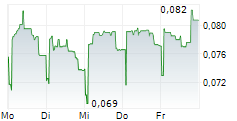
GENSIGHT BIOLOGICS SA gehört der Branche Biotechnologie an. Der aktuelle Kurs liegt bei 0,081 Euro und damit 0,00 % im Plus.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu GENSIGHT BIOLOGICS SA finden Sie auf Wallstreet Online.